Summary
-
1.
Solubilities of nitrites of sodium, potassium and silver have been determined in anhydrous ethylenediamine and monoethanolamine.
-
2.
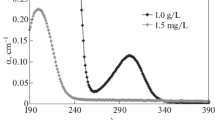
Silver nitrite forms a crystalline solvate with ethylenediamine with this formula AgNO2-2 [C2H4(NH2)2].
-
3.
The ethylenediamine solutions of silver and sodium nitrites are more conducting than those of monoethanolamine; however, potassium nitrite behaves contrary to the above trend.
-
4.
Sodium nitrite shows a conductance minimum in ethylenediamine but not in monoethanolamine. Such behaviour has been encountered in ethylenediamine with mercuric cyanide13.
Similar content being viewed by others
References
For Part II, see Muniyappan, T. and Anjaneyalu, B.Proc. Ind. Acad. Sci., 1957,45 A, 412.
Das, Palit and Somayajulu “Non-AqueousTitration,”Indian Association for Cultivation of Science, Calcutta, 1954, p. 26.
Drake, N. L. and Plummer, C. W.J. Am. Chem. Soc., 1954,76, 2720.
Isbin, H. and Kobe, K. A. —, 1945,67, 464.
Franklin and KrausAm. Chem. J., 1898,20, 824.
Mellor, J. W.Comprehensive Treatise on Inorganic and Theoretical Chemistry, 1928,8, 483.
Moeller, T. and Zimmermann, P.A.J. Am. Chem. Soc., 1953,75, 3940.
Bromley, W. H. and Luder, W. F. —, 1944,66, 107.
Hibbard, B. B. and Schmidt, F. C. —, 1955,77, 225.
Fuoss, R. M. and Kraus, C. A. —, 1933,55, 2387.
Kraus, CA.J. Phys. Chem., 1956,60, 129–141.
Huckel, W.Structural Chemistry of Inorganic Compounds, 1950,1, 131.
Davis, R. E., Peacock, J., Schaap, W. B. and Schmidt, F. C.J. Am. Chem. Soc., 1955,77, 5831.
Author information
Authors and Affiliations
Additional information
Communicated by Dr. T. R. Govindachari,f.a.sc.
Rights and permissions
About this article
Cite this article
Muniyappan, T., Anjaneyalu, B. Investigations in non-aqueous solvents. Proc. Indian Acad. Sci. 46, 454–461 (1957). https://doi.org/10.1007/BF03045958
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03045958