Summary
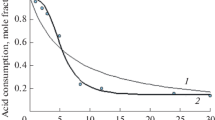
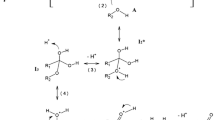
In continuation of our studies on ester hydrolysis, the kinetics of alkaline hydrolysis of succinates are reported. The rate constants of the first and second step of hydrolysis have been evaluated. A comparative study of the structural and solvent influences on the succinates is made.
Similar content being viewed by others
References
Frost and Schwemer..J. Amer. Chem. Soc., 1952,74, 1268.
Meyer..Z. Physik. Chem., 1909,67, 257.
Branch and Calvin..Theory of Organic Chemistry, Prentice Hall, New York, 1946.
Ingold ..J. Chem. Soc., 1930, 1032.
Svirbely..J. Amer. Chem. Soc., 1959,81, 255.
Hammett.. Ibid., 1958,80, 2415.
----- .. Private Communication.
Ritchie ..J. Chem. Soc., 1931, 3112.
Westheimer..J. Chem. Phys., 1942,10, 478.
Author information
Authors and Affiliations
Additional information
Communicated by Prof. S. V. Anantakrishnan,f.a.sc.
Rights and permissions
About this article
Cite this article
Radhakrishnamurti, P.S. Kinetic studies in ester hydrolysis. Proc. Indian Acad. Sci. 54, 325–330 (1961). https://doi.org/10.1007/BF03045851
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03045851