Summary
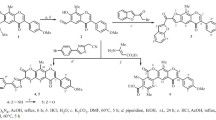
7-Allyloxyflavone undergoes Claisen transformation to form 8-allyl-7-hydroxyflavone, the constitution of which is established from its synthesis from 3-allyl-resacetophenone. The bond distribution should therefore be as in (I). However 8-alkyl substituted derivatives undergo coupling with diazotisedp-nitraniline to form dyes and 7-allyloxy-8-allylflavone undergoes further transformation to form 6: 8-diallyl-7-hydroxy-flavone. Closely analogous results are obtained from 2-methyl-3-methoxy-7-hydroxychromone. It is therefore concluded that though the chromones generally reat in form (I) the alternative disposition of the bonds (IV) is not precluded.
Similar content being viewed by others
References
Wittig, Baugert and ReichertAnn., 1925,184.
Baker, W.J.C.S., 1934,73.
Baker, W. and (Miss) LothianIbid., J.C.S., 1935,628.
Rangaswami, S., and Seshadri, T. R.Proc. Ind. Acad. Sci., (A), 1938,8, 214.
Allan, J., and Robinson, R.,J.C.S., 1924,2192.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rangaswami, S., Seshadri, T.R. Fixation of the aromatic double bonds in the chromones. Proc. Indian Acad. Sci. (Math. Sci.) 9, 1–6 (1939). https://doi.org/10.1007/BF03045441
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03045441