Summary
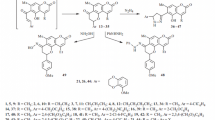
A general method for the preparation of 5-hydroxyflavone and its derivatives from the corresponding 5:7-dihydroxyflavones by the action of Raney nickel on the 7-tosyloxy derivatives is described. Thus chrysin has been converted into 5-hydroxyflavone, and galangin 3-methyl ether to 5-hydroxy-3-methoxyflavone.
Similar content being viewed by others
References
Karrer and Schwab..Helv. Chim. Acta, 1941,24, 297.
Sugasawa ..J. Chem. Soc., 1934, 1483.
Baker ..Ibid., 1934, 1953.
See also Rajagopalan, Rao and SeshadriProc. Ind. Acad. Sci., 1947,25A, 432.
Iyer and Venkataraman.., 1953,37A, 629.
Seshadri and Venkateswarlu.., 1947,26A, 189.
Kenner and Murray ..J. Chem. Soc., 1949, S178.
Ramanathan and Venkataraman..Curr. Sci., 1952,21, 283.
Robinson and Venkataraman ..J. Chem. Soc., 1926, 2344.
Rao, Rao and Seshadri..Proc. Ind. Acad. Sci., 1947,25A, 427.
Kalff and Robinson..J. Chem. Soc., 1925,127, 181.
Wilson..J. Am. Chem. Soc., 1939,61, 2304.
Mahal, Rai and Venkataraman ..J. Chem. Soc., 1935, 866.
Karmarkar, Shah and VenkataramanProc. Ind. Acad. Sci., 1952,36A, 552.
Seshadri, Varadarajan and Venkateswarlu, 1950,32A, 250.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ramanathan, V., Venkataraman, K. Synthetical experiments in the chromone group. Proc. Indian Acad. Sci. (Math. Sci.) 38, 40–44 (1953). https://doi.org/10.1007/BF03045234
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03045234