Abstract
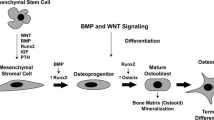
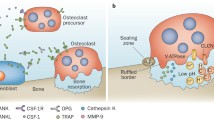
Bone resorption in a common feature of rheumatoid and other inflammatory joint conditions. This osteolysis is effected by osteoclasts, highly specialised multinucleated cells which are formed by fusion of mononuclear phagocyte precursors of haematopoietic origin. It has been shown that human osteoclast precursors circulate in the monocyte fraction of peripheral blood and are present in the macrophage population of normal and pathological tissues. The cellular and humoral requirements for human monocyte/macrophage-osteoclast differentiation are the presence of bone-derived stromal cells, which express the membrane-bound protein termed osteoclast differentiation factor or osteoprotegerin ligand (ODF/OPGL), and macrophage-colony stimulating factor (M-CSF). Macrophages form a major component of the inflammatory infiltrate in the rheumatoid synovium and in other conditions such as PVNS and aseptic loosening. Local factors (e.g. cytokines and growth factors) produced in this microenvironment act to stimulate not only osteoclast activity but also osteoclast formation. In the context of RA and other joint conditions in which osteolysis occurs, pathological bone resorption should be regarded as a function not only of osteoclast activity but also of osteoclast formation from mononuclear phagocyte precursors.
Similar content being viewed by others
References
Langevoort HL, Cohn ZA, Hirsch JGet al., The nomenclature of mononuclear phagocytic cells — proposal for a new classification, in:Mononuclear Phagocytes, van Furth R (Ed.), pp. 1–6. Blackwell Scientific Publications, Oxford (1970).
van Furth R, The mononuclear phagocyte system: a new classification of macrophages, monocytes and their precursor cells,Bull WHO 46, 845–852 (1972).
Groopman JE, Golde DW, The histiocytic disorders. A pathophysiologic analysis,Ann Intern Med 94, 95–107 (1981).
van Furth R (Ed.),Mononuclear Phagocytes: Functional Aspects. Nijhoff, Dordrecht (1980).
Ghadially FN, The articular territory of the reticulo-endothelial system,Ultrastruct Pathol 1, 249–264 (1980).
Burmester GR, Loacher P, Winchester RJet al., The tissue architecture of synovial membranes in inflammatory and non inflammatory joint disease,Rheumatol Int 3, 173–181 (1983).
Hogg N, Palmer DG, Revell PA, Mononuclear phagocytes of normal and rheumatoid synovial membrane identified by monoclonal antibodies,Immunology 56, 673–681 (1985).
Athanasou NA, Quinn J, Heryet Aet al., The immunohistology of synovial lining cells in normal and inflamed synovium,J Pathol 155, 133–142 (1988).
Henderson B, Revell PA, Edwards JC, Synovial lining cell hyperplasia in rheumatoid arthritis: dogma and fact,Ann Rheum Dis 47, 348–349 (1988).
Allen CA, Highton J, Palmer GD, Increased expression of P150, 95 and CR3 leukocyte adhesion molecules by mononuclear phagocytes in rheumatoid synovial membrane. Comparison with osteoarthritis and normal synovial membranes,Arthritis Rheum 32, 947–954 (1989).
Hale LP, Martin ME, McCollum DEet al., Immunohistologic analysis of the distribution of cell adhesion molecules within the inflammatory synovial microenvironment,Arthritis Rheum 32, 22–30 (1989).
Broker BM, Edwards JCW, Fanger MWet al., The prevalence and the distribution of macrophage bearing FcRI, FcRII and FcRIII in synovium,Scand J Rheumatol 19, 123–135 (1990).
Athanasou NA, Immunophenotypic analysis of mononuclear phagocyte antigens in the arthritic synovium of the hip joint,Hip Int 3, 67–74 (1993).
Ziff M, Pathways of mononuclear cell infiltration in rheumatoid synovitis,Rheumatol Int 9, 97–103 (1989).
Athanasou NA, The cellular biology of bone resorbing cells,J Bone Joint Surg (Review) 78A, 1096–1112 (1996).
Gothlin G, Ericsson JLE, The osteoclast: review of ultrastructure, origin, and structure-function relationship,Clin Orhop 120, 201–231 (1976).
Tonna EA, Cronkite EP, Use of tritiated thymidine for the study of the origin of the osteoclast,Nature 190, 459–460 (1961).
Walker DG, Osteopetrosis cured by temporary parabiosis,Science 180, 875 (1973).
Gothlin G, Ericsson JL, On the histogenesis of the cells in fracture callus. Electron microscopic autoradiographic observations in parabiotic rats and studies on labeled monocytes,Virchows Arch [B] Cell Pathol 12, 318–329 (1973).
Kahn AJ, Simmons DJ, Investigation of cell lineage in bone using a chimaera of chick and quial embryonic tissue,Nature 258, 325–327 (1975).
Walker DG, Bone resorption restored in osteopetrotic mice by transplants of normal bone marrow and spleen cells,Science 190, 784–785 (1975).
Walker DG, Control of bone resorption by hematopoietic tissue. The induction and reversal of congenital osteopetrosis in mice through use of bone marrow and splenic transplants,J Exp Med 142, 651–663 (1975).
Coccia PF, Krivit W, Cervenka Jet al., Successful bone-marrow transplantation for infantile malignant osteopetrosis,N Engl J Med 302, 701–708 (1980).
Burger EH, van der Meer JW, van de Gevel JSet al., In vitro formation of osteoclasts from longterm cultures of bone marrow mononuclear phagocytes,J Exp Med 156, 1604–1614 (1982).
Ibbotson KJ, Roodman GD, McManus LMet al., Identification and characterization of osteoclast-like cells and their progenitors in cultures of feline marrow mononuclear cells,J Cell Biol 99, 471–480 (1984).
Kurihara N, Chenu C, Miller Met al., Identification of committed mononuclear precursors for osteoclast-like cells formed in long term human marrow cultures,Endocrinology 126, 2733–2741 (1990).
Kurihara N, Gluck S, Roodman GD, Sequential expression of phenotype markers for osteoclasts during differentiation of precursors for multinucleated cells formed in long-term human marrow cultures,Endocrinology 127, 3215–3221 (1990).
Sarma U, Flanagan AM, Macrophage colony-stimulating factor induces substantial osteoclast generation and bone resorption in human bone marrow cultures,Blood 88, 2531–2540 (1996).
Takahashi N, Yamana H, Yoshiki Set al., Osteoclast-like cell formation and its regulation by osteotropic hormones in mouse bone marrow cultures,Endocrinology 122, 1373–1382 (1988).
Udagawa N, Takahashi N, Akatsu Tet al., Origin of the osteoclast: mature monocytes and macrophages are capable of differentiating into osteoclasts under a suitable microenvironment prepared by bone marrow derived cells,Proc Natl Acad Sci USA 87, 7260–7264 (1990).
Quinn JM, McGee JO, Athanasou NAet al., Cellular and hormonal factors influencing monocyte differentiation to osteoclastic bone-resorbing cells,Endocrinology 134, 2416–2423 (1994).
Quinn J, Joyner C, Triffitt JTet al., Polymethylmethacrylate-induced inflammatory macrophages resorb bone,J Bone Joint Surg 74(B), 652–658 (1992).
Quinn JM, Athanasou NA, Tumour infiltrating macrophages are capable of bone resorption,J Cell Sci 101, 681–686 (1992).
Quinn JM, Matsumura Y, Tarin Det al., Cellular and hormonal mechanisms associated with malignant bone resorption,Lab Invest 71, 465–471 (1994).
Shin JH, Kukita A, Ohki Ket al., In vitro differentiation of the murine macrophage cell line BDM-1 into osteoclast-like cells,Endocrinology 136, 4285–4292 (1995).
Quinn JM, Sabokbar A, Athanasou NA, Cells of the mononuclear phagocyte series differentiate into osteoclastic lacunar bone resorbing cells,J Pathol 179, 106–111 (1996).
Athanasou NA, Quinn JM, Human tumour-associated macrophages are capable of bone resorption,Br J Cancer 65, 523–526 (1992).
Fujikawa Y, Quinn JM, Sabokbar Aet al., The human osteoclast precursor circulates in the monocyte fraction,Endocrinology 137, 4058–4060 (1996).
Roussel MF, Downing JR, Rettenmeier CWet al., A point mutation in the extracellular domain of the human CSF-1 receptor (c-fms product) activates its transforming potential,Cell 55, 979–984 (1988).
Fujikawa Y, Sabokbar A, Neale Set al., Human osteoclast formation and bone resorption by monocytes and synovial macrophages in rheumatoid arthritis,Ann Rheum Dis 55, 816–822 (1996).
Sabokbar A, Fujikawa Y, Neale Set al., Human arthroplasty derived macrophages differentiate into osteoclastic bone resorbing cells,Ann Rheum Dis 56, 414–420 (1997).
Quinn JM, Fujikawa Y, McGee JOet al., Rodent osteoblast-like cells support osteoclastic differentiation of human cord blood monocytes in the presence of M-CSF and 1,25 dihydroxyvitamin D3,Int J Biochem Cell Biol 29, 173–179 (1997).
Quinn JM, Neale S, Fujikawa Yet al.: Human osteoclast formation from blood monocytes, peritoneal macrophages, and bone marrow cells,Calcif Tissue Int (1998) (in press).
Takahashi N, Akatsu T, Udagawa Net al., Osteoblastic cells are involved in osteoclast formation,Endocrinology 123, 2600–2602 (1988).
Udagawa N, Takahashi N, Akatsu Tet al., The bone marrow-derived stromal cell lines MC3T3-G2/PA6 and ST2 support osteoclast-like cell differentiation in co-cultures with mouse spleen cells,Endocrinology 125, 1805–1813 (1989).
Hiura K, Sumitani K, Kawata Tet al., Mouse osteoblastic cells (MC3T3-E1) at different stages of differentiation have opposite effects on osteoclastic cell formation,Endocrinology 128, 1630–1637 (1991).
Tanaka S, Takahashi N, Akatsu Tet al., Macrophage colony stimulating factor is indispensible for both proliferation and differentiation of osteoclast progenitors,J Clin Invest 91, 257–263 (1993).
Yoshida H, Hayashi S-I, Kunisada Tet al., The murine mutation osteopetrosis is in the coding region of the macrophage colony stimulating factor gene,Nature 345, 442–444 (1990).
Felix R, Cecchini MG, Fleisch H, Macrophage colony stimulating factor restoresin vivo bone resorption in theop/op osteopetrotic mouse,Endocrinology 127, 2592–2594 (1990).
Felix R, Cecchini MG, Hofstetter Wet al., Impairment of macrophage colony-stimulating factor production and lack of resident bone marrow macrophage in the osteopetroticop/op mouse,J Bone Miner Res 5, 781–789 (1990).
Kodama H, Nose M, Niida Set al., Essential role of macrophage colony-stimulating factor in the osteoclast differentiation supported by stromal cells,J Exp Med 173, 1291–1294 (1991).
Suda T, Takahashi N, Martin TJ, Modulation of osteoclast differentiation,Endocrine Reviews 13, 66–80 (1992).
Suda T, Udagawa N, Nakamura Iet al., Modulation of Osteoclast differentiation by local factors,Bone 17, 87S-91S (1995).
Matayoshi A, Brown C, DiPersio JFet al., Human blood-mobilized hematopoietic precursors differentiate into osteoclasts in the absence of stromal cells,Proc Natl Acad Sci USA 93, 10785–10790 (1996).
Higuchi S, Tabata N, Tajima Met al., Induction of human osteoclast-like cells by treatment of blood monocytes with anti-fusion regulatory protein-1/CD98 monoclonal antibodies,J Bone Miner Res 13, 44–49 (1998).
Simonet WS, Lacey DL, Dunstan CRet al., Osteoprotegerin: a novel secreted protein involved in the regulation of bone density,Cell 89, 309–319 (1997).
Tsuda E, Goto M, Mochizuki Set al., Isolation of a novel cytokine from human fibroblasts that specifically inhibits osteoclastogenesis,Biochem Biophys Res Commun 234, 137–142 (1997).
Yasuda H, Shima N, Nakagawa Net al., Identity of osteoclastogenesis inhibitory factor (OCIF) and osteoprotegerin (OPG): a mechanism by which OPG/OCIF inhibits osteoclastogenesisin vitro, Endocrinology 139, 1329–1337 (1998).
Yasuda H, Shima N, Nakagawa Net al., Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL,Proc Natl Acad Sci USA 95, 3597–3602 (1998).
Lacey DL, Timms E, Tan HLet al., Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation,Cell 93, 165–176 (1998).
Bromley M, Woolley DE, Chondroclasts and osteoclasts at subchondral sites of erosion in the rheumatoid joint,Arthritis Rheum 27, 968–975 (1984).
Yanni G, Whelan A, Feighery Cet al., Synovial tissue macrophages and joint erosion in rheumatoid arthritis,Ann Rheum Dis 53, 39–44 (1994).
Chu CQ, Field M, Allard Set al., Detection of cytokines at the cartilage/pannus junction in patients with rheumatoid arthritis: implications for the role of cytokines in cartilage destruction and repair,Br J Rheumatol 31, 653–661 (1992).
Zvaifler NJ, Firestein GS, Pannus and pannocytes. Alternative models of joint destruction in rheumatoid arthritis,Arthritis Rheum 37, 783–789 (1994).
Tetlow LC, Lees M, Woolley DE, Comparative studies of collagenase and stromelysin-1 expression by rheumatoid synoviocytesin vitro, Virchows Arch 425, 569–576 (1995).
Jensen HS, Jensen LT, Saxne Tet al., Human monocyte elastolytic activity, the propeptides of types I and III procollagen, proteoglycans, and interleukin-6 in synovial fluid from patients with arthritis,Clin Exp Rheumatol 9, 391–394 (1991).
Bromley M, Woolley DE, Histopathology of the rheumatoid lesion. Identification of cell types at sites of cartilage erosion,Arthritis Rheum 27, 857–863 (1984).
Chang JS, Quinn JM, Demaziere Aet al., Bone resorption by cells isolated from rheumatoid synovium,Ann Rheum Dis 51, 1223–1229 (1992).
Wilkinson LS, Pitsillides AA, Edwards JC, Giant cells in arthritic synovium,Ann Rheum Dis 52, 182–184 (1993).
Ashton BA, Ashton IK, Marshall MJet al., Localisation of vitronectin receptor immunoreactivity and tartrate resistant acid phosphatase activity in synovium from patients with inflammatory of degenerative arthritis,Ann Rheum Dis 52, 133–137 (1993).
Kotake S, Sato K, Kim KJet al., Interleukin-6 and soluble interleukin-6 receptors in the synovial fluids from rheumatoid arthritis patients are responsible for osteoclast-like cell formation,J Bone Miner Res 11, 88–95 (1996).
Fujikawa Y, Shingu M, Torisu Tet al., Bone resorption by tartrate-resistant acid phosphatase-positive multinuclear cells isolated from rheumatoid synovium,Br J Rheumatol 35, 213–217 (1996).
Takayanagi H, Oda H, Yamamoto Set al., A new mechanism of bone destruction in rheumatoid arthritis: synovial fibroblasts induce osteoclastogenesis,Biochem Biophys Res Commun 240, 279–286 (1997).
Hayes ME, Denton J, Freemont AJet al., Synthesis of the active metabolite of vitamin D, 1,25(OH)2D3, by synovial fluid macrophages in arthritic diseases,Ann Rheum Dis 48, 723–729 (1989).
Seitz M, Hunstein W, Enhanced prostanoid release from monocytes of patients with rheumatoid arthritis and active systemic lupus erythematosus,Ann Rheum Dis 44, 438–445 (1985).
Seitz M, Loetscher P, Fey MFet al., Constitutive mRNA and protein production of macrophage colony-stimulating factor but not of other cytokines by synovial fibroblasts from rheumatoid arthritis and osteoarthritis patients,Br J Rheumatol 33, 613–619 (1994).
Fujii I, Shingu M, Noburaga M, Monocyte activation in early onset of rheumatoid arthritis,Ann Rheum Dis 49, 497–503 (1990).
Gardner DL,Pathological Basis of the Connective Tissue Diseases. Edward Arnold, London (1992).
Bullough PG, DiCarlo EF, Hansraj KKet al., Pathologic studies of total joint replacement,Orthop Clin North Am 19, 611–625 (1988).
Lennox DW, Schofield BW, McDonald DFet al., A histologic comparison of aseptic loosening of cemented, press-fit and biologic ingrowth prostheses,Clin Orthop 225, 171–191 (1987).
Revell PA, Tissue reactions to joint prostheses and the products of wear and corrosion, in:Current Trends in Pathology, Berry C (Ed.), pp. 73–101. Springer-Verlag, Berlin (1982).
Quinn JMW, Joyner C, Triffitt JTet al., Polymethylmethacrylate-induced inflammatory macrophages resorb bone,J Bone Joint Surg 72B, 625–628 (1992).
Pandey R, Quinn J, Joyner Cet al., Arthroplasty implant biomaterial-associated macrophages differentiate into lacunar bone-resorbing cells,Ann Rheum Dis 55, 388–395 (1996).
Sabokbar A, Pandey R, Quinn JMet al., Osteoclastic differentiation by mononuclear phagocytes containry biomaterial particles,Acta Ortho Trauma Surg 11, 136–140 (1998).
Horowitz SM, Purdon MA, Mechanisms of cellular recruiment in aseptic loosening of prosthetic joint implants,Calcif Tissue Int 57, 301–305 (1995).
Glant TT, Jacobs JJ, Response of three murine macrophage populations to particulate debris: bone resorption in organ cultures,J Orthop Res 12, 720–731 (1994).
Haynes DR, Rogers SD, Hay Set al., The differences in toxicity and release of bone-resorbing mediators induced by titanium and cobalt-chromium-alloy wear particles,J Bone Joint Surg 75A, 825–834 (1993).
Horowitz SM, Doty SB, Lane JMet al., Studies of the mechanism by which the mechanical failure of polymethylmethacrylate leads to bone resorption,J Bone Joint Surg 75A, 802–813 (1993).
Shanbhag AS, Jacobs JJ, Black Jet al., Human monocyte response to particulate biomaterials generatedin vivo andin vitro, J Orthop Res 13, 792–801 (1995).
Blaine TA, Rosier RN, Puzas JEet al., Increased levels of tumor necrosis factor-A and interleukin-6 protein and messenger RNA in human peripheral blood monocytes due to titanium particles,J Bone Joint Surg 78A, 1181–1192 (1996).
Quinn JMW, McGee JO, Athanasou NA, Human tumour-associated macrophages differentiate into osteoclastic bone resorbing cells,J Pathol 184, 31–36 (1998).
Neale SD, Kristelly R, Gundle Ret al., Giant cells in pigmented villo nodular synovitis express an osteoclast phenotype,J Clin Pathol 50, 605–608 (1997).
Athanasou NA, Puddle B, Quinn Jet al., Use of monoclonal antibodies to recognise osteoclasts in routinely processed bone biopsy specimens,J Clin Pathol 44, 664–666 (1991).
Quinn JMW, Sabokbar A, Denne Met al., Inhibitory and stimulatory effects of prostaglandins on osteoclast differentiation,Calcif Tissue Int 60, 63–70 (1997).
Author information
Authors and Affiliations
About this article
Cite this article
Fujikawa, Y., Athanasou, N.A. Pathological bone resorption in rheumatic diseases. Japanese Journal of Rheumatology 8, 355–367 (1998). https://doi.org/10.1007/BF03041315
Issue Date:
DOI: https://doi.org/10.1007/BF03041315