Abstract
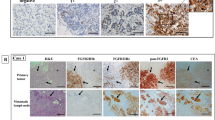
To assess the significance of epidermal growth factor receptor family members, the overexpression of c-erbB-1 and c-erbB-2 was retrospectively investigated in 146 southern Iranian gastric cancer patients. Indirect immunostaining was used to evaluate the expression of these two receptors in formalin-fixed paraffin-embedded tissue samples. c-ErbB-1 expression was observed in 47 (32.2%) and c-erbB-2 expression was observed in 24 (16.4%) of tumors. Significant positive correlations were observed between c-erbB-1 expression and tumor size, local invasion, lymph node involvement and tumor stage. There was also a negative correlation between c-erbB-2 expression and tumor stage. These results may suggest the contribution of c-erbB-1 molecule in progression of gastric carcinomas in southern Iranian patients. Moreover, the relatively high percentage of c-erbB-2 positive tumors may provide a useful target for the immunotherapy of these cancers.
Similar content being viewed by others
References
Farahmandbeigi M, Kadivar MR: The incidence rate of registered cancers in Fars province. Disease Control Unit, Shiraz University Press, Iran, 2000.
Koyama S, Maruyama T, Adachi S: Expression of epidermal growth factor receptor and CD44 splicing variants sharing exons 6 and 9 on gastric and esophageal carcinomas: a twocolor flow-cytometric analysis. J Cancer Res Clin Oncol 125: 47–54, 1999.
Prakash I, Mathur RP, Kar P, et al: Comparative evaluation of cell proliferative indices and epidermal growth factor receptor expression in gastric carcinoma. Indi J Pathol Microbiol 40:481–490, 1997.
Jang WI, Yang WI, Lee CI, et al: Immunohistochemical detection of p53 protein, c-erbB-2 protein, epidermal growth factor receptor protein and proliferating cell nuclear antigen in gastric carcinoma. J Korean Med Sci 8: 293–304, 1993.
Oshima CT, Lanzoni VP, Iriya K, Forones NM: C-erbB-2 oncoprotein in gastric carcinoma: correlation with clinical stage and prognosis. Int J Biol Markers 16: 250–254, 2001.
Wu MS, Shun CT, Sheu JC, et al: Overexpression of mutant p53 and c-erbB-2 proteins and mutations of the p15 and p16 genes in human gastric carcinoma: with respect to histological subtypes and stages. J Gastroenterol Hepatol 13: 305–310, 1998.
Lee HR, Kim JH, Uhm HD, et al: Overexpression of c-ErbB-2 protein in gastric cancer by immunohistochemical stain. Oncology 53: 192–197, 1996.
Aoyagi K, Kohfuji K, Yano S, et al: Evaluation of the epidermal growth factor receptor (EGFR) and c-erbB-2 in superspreading-type and penetrating-type gastric carcinoma. Kurume Med J 48:197–200, 2001.
Motojima K, Furui J, Kohara N, et al: erbB-2 expression in welldifferentiated adenocarcinoma of the stomach predicts shorter survival after curative resection. Surgery 115:349–354, 1994.
Yoshida K, Yasui W, Ito H, Tahara E: Growth factors in progression of human esophageal and gastric carcinomas. Exp Pathol 40: 291–300, 1990.
Lee EY, Cibull ML, Strodel WE, Haley JV: Expression of HER-2/neu oncoprotein and epidermal growth factor receptor and prognosis in gastric carcinoma. Arch Pathol Lab Med 118: 235–239, 1994.
Orita H, Maehara Y, Emi Y, et al: c-erbB-2 expression is predictive for lymphatic spread of clinical gastric carcinoma. Hepatogastroenterology 44: 294–298, 1997.
Tokunaga A, Onda M, Okuda T, et al: Prevention of growth of a human gastric cancer xenograft in nude mice with anti-EGF receptor ntibody. In: Takahashi T, editor. Recent advances in management of digestive cancers. Tokyo: Springer-Verlag, 403–405, 1993.
Teramoto T, Onda M, Tokunaga A, Asano G: Inhibitory effect of anti-epidermal growth factor receptor antibody on a human gastric cancer. Cancer 77s: 1639–1645, 1996.
Ross JS, McKenna BJ: The HER-2/neu oncogene in tumors of the gastrointestinal tract. Cancer Invest 19:554–568, 2001.
Modjtahedi H, Hickish T, Nicolson M, et al: Phase I trial and tumour localisation of the anti-EGFR monoclonal antibody ICR62 in head and neck or lung cancer. Br J Cancer 73: 228–235, 1996.
Bianco C, Bianco R, Tortora G, et al: Antitumor activity of combined treatment of human cancer cells with ionizing radiation and anti-epidermal growth factor receptor monoclonal antibody C225 plus type I protein kinase A antisense oligonucleotide. Clin Cancer Res 6: 4343–4350, 2000.
Ciardiello F, Bianco R, Damiano V, et al: Antiangiogenic and antitumor activity of anti-epidermal growth factor receptor C225 monoclonal antibody in combination with vascular endothelial growth factor antisense oligonucleotide in human GEO colon cancer cells. Clin Cancer Res 6: 3739–47, 2000.
Ciardiello F, Bianco R, Damiano V, et al: Antitumor activity of sequential treatment with topotecan and anti-epidermal growth factor receptor monoclonal antibody C225. Clin Cancer Res 5: 909–916, 1999.
Falcey J, Pfister D, Cohen R, et al: A study of anti-epidermal growth factor receptor (EGFr) monoclonal antibody C225 and cisplastin in patients with head and neck or lung carcinomas. Proc Am Soc Clin Oncol 16: 1364, 1997.
Cobleigh MA, Vogel CL, Tripathy D, et al: Multinational study of the efficacy and safety of humanized anti-HER2 monoclonal antibody in women who have HER2-overexpressing metastatic breast cancer that has progressed after chemotherapy for metastatic disease. J Clin Oncol 17: 2639–2648, 1999.
Slamon DJ, Leyland-Jones B, Shak S, et al: Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344: 783–792, 2001.
Vogel CL, Cobleigh MA, Tripathy D, et al: Efficacy and safety of trastuzumab as a single agent in first-line treatment of HER2-overexpressing metastatic breast cancer. J Clin Oncol 20: 719–726, 2002.
Cook-Bruns N: Retrospective analysis of the safety of Herceptin immunotherapy in metastatic breast cancer. Oncology 61s: 58–66, 2001.
Azzoli CG, Krug LM, Miller VA, et al: Trastuzumab in the treatment of non-small cell lung cancer. Semin Oncol 29s: 59–65, 2002.
Zinner RG, Kim J, Herbst RS: Non-small cell lung cancer clinical trials with trastuzumab: their foundation and preliminary results. Lung Cancer 37: 17–27, 2002.
Leyland-Jones B: Maximizing the response to Herceptin therapy through optimal use and patient selection. Anticancer Drugs 4s: 11–17, 2001.
Modjtahedi H, Eccles SA, Box G, et al: Antitumor activity of combinations of antibodies directed against different epitopes on the extracellular domain of the human EGF receptor. Cell Biophys 22: 129–146, 1993.
Styles JM, Harrison S, Gusterson BA, Dean CJ: Rat monoclonal antibodies to the external domain of the product of the C-erbB-2 proto-oncogene. Int J Cancer 45: 320–324, 1990.
Walker RA: The erbB/HER type 1 tyrosine kinase receptor family. J Pathol 185: 234–235, 1998.
Peles E, Yarden Y: Neu and its ligands: from an oncogene to neural factors. Bioessays 15: 815–824, 1993.
Takehana T, Kunitomo K, Kono K, et al: Status of c-erbB-2 in gastric adenocarcinoma: a comparative study of immunohistochemistry, fluorescence in situ hybridization and enzymelinked immuno-sorbent assay. Int J Cancer 98: 833–837, 2002.
Gharesi-Fard B, Vasei M, Talei A, et al: The expression and prognostic significance of c-erbB-2 molecules in patients with breast cancer in Iran. Irn J Med Sci 25: 31–35, 2000.
Khademi B, Shirazi FM, Vasei M, et al: The expression of p53, c-erbB-1 and c-erbB-2 molecules and their correlation with prognostic markers in patients with head and neck tumors. Cancer Lett 184: 223–230, 2002.
Shimada E, Kato M, Saito Y: Immunohistochemical study of the c-erbB-2 protein in human gastric carcinoma. Nippon Geka Gakkai Zasshi 94: 33–40, 1993.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghaderi, A., Vasei, M., Maleck-Hosseini, S. et al. The expression of c-erbB-1 and c-erbB-2 in Iranian patients with gastric carcinoma. Pathol. Oncol. Res. 8, 252–256 (2002). https://doi.org/10.1007/BF03036740
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03036740