Abstract
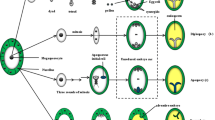
The reproductive system determines the way in which gametes develop and interact to form a new organism. Therefore, it exerts the primary level of control of genotypic frequencies in plant populations, and plays a fundamental role in plant breeding. A basic understanding of plant reproductive development will completely transform current breeding strategies used for seed production. Apomixis is an asexual form of reproduction in which embryogenesis occurs in a cell lineage lacking both meiosis and fertilization, and that culminates in the formation of viable progeny genetically identical to the mother plant. The transfer of apomixis into sexual crops will allow the production of self-perpetuating improved hybrids, and the fixation of any desired heterozygous genotype. The initiation of apomictic development invariably takes place at early stages of ovule ontogeny, before the establishment of the megagametophytic phase. The developmental versatility associated with megagametophyte formation suggests that the genetic and molecular regulation of apomixis is intimately related to the regulation of sexuality. Differences between the initiation of sexual and apomictic development may be determined by regulatory genes that act during megasporogenesis, and that control events leading to the formation of unreduced female gametophytes. To test this hypothesis, we are isolating and characterizing genes that act during megasporogenesis inArabidopsis thaliana and investigating their potential role in the induction of apomixis. We are using a recently established transposon-based enhancer detection and gene trap insertional mutagenesis system that allows the identification of genes based on their expression patterns. An initial screen of transposants has yielded over 20 lines conferring restricted GUS expression during early ovule development. We have obtained the sequence of genomic fragments flanking the transposon insertion. Several have homology to genes playing important roles in plant and animal development. They include cell cycle regulators, enzymes involved in callose hydrolysis, leucine-rich repeat protein kinase receptors, and expressed sequence tags (ESTs) of unknown function. Independently, a genetic screen allows the identification of female sterile mutants defective in megasporogenesis. Results from these experiments will improve our basic understanding of reproductive development in plants, and will set the basis for a sustained effort in plant germ line biotechnology, a first step toward a flexible transfer of apomixis into a large variety of sexual crops.
Similar content being viewed by others
Literature Cited
Asker, S.E. and L. Jerling. 1992. Apomixis in plants. Boca Raton: CRC Press.
Baker, U.S., A.T.C. Cartpenter, M.S. Esposito, R.E. Esposito and E. Sandler. 1976. The genetic control of meiosis.Ann. Rev. den.10: 53–134.
Bell, P.R. 1992. Aposporv and apogamy: implications for understanding the plant life cyclc.Int. J. Plant Sci.153: 123–136.
Bellen, H.J., C.J. O’Kane, C. Wilson, U. Grossniklaus, R.K. Pearson and W.J. Gehring. 1989. P-element mediated enhancer detection: a versatile method to study development inDrosopltila.Genes and Dev.3: 1288–1300.
Bier, E., H. Vaessin, S. Shepherd, K. Fee, K. McCall, S. Barbel, L. Ackerman, R. Carretto, T. Uemura, E. Grell, L.Y. Jan and V.N. Jan. 1989. Searching for pattern and mutation in theDrosophila genome with P-lacZ vector,Genes and Dev.3. 1273–1287.
Bicknell, R.A. 1997. Isolation of a diploid, apomictic plant ofHieraciatn aurantiaeam.Sex. Plant Reprod.10: 168–172.
Bouchard, R.A. 1990. Characterisation of expressed inactive transcript clones of Lilium, relatedness, and affinity to small heal shock proteins,Genome33: 68–76.
Bretagnolle, F. and J.D. Thompson. 1995. Gametes with the somatic chromosome number: mechanisms of then formation and role in the evolution of autopoly-ploid plants.New Phviol.129: 1–22.
Burton, G.W. 1992. Manipulating apomixis inPaspatum. In Proceedings of the Apomixis Workshop. USDA-ARS. Atlanta, pp 16–14.
Cass, D.D. and W.A. Jensen. 1970. Fertilization in barky.Amer. J. Hot.57: 62–70.
Chaudhry, A.M., L. Ming, C. Miller, S. Craig, E.S. Dennis and W.J. Peacock. 1997. Fertilization-independent seed development inArabidopsis thaliana.Proc. Nat. Acad. Sci. USA15: 4223–4228.
Christensen, C.A., E.J. King, J.R. Jordan and G.N. Drews. 1667. Megagametogenesis in Arabidopsis wild type and Gf mutant.Sex. Plant Reprod.10: 46–64.
Coen, E.S. and E. Meverowilz 1661. The war of the whorls.Nature353: 31–37
Curie, C. and S. McCormick. 1997. A strong inhibitor of gene expression in the 5’ untranslated region of the pollen-specific LAT59 gene totomato.The Plant Cell9: 2025–2036.
D’Amatom, F. 1989. Polyploidy in cell differentiation.Carvologis42: 183–211.
De Haan, A., N.O. Maceira, R. Lumaret and J. Delay 1992. Production of 2n gametes in diploid subspecies ofDactxlis glomerata L. 2. Occurrence and Ircqueney of 2n eggs.Ann. of Bat.69: 345–350.
Dickinson, H.G. 1995. The regulation of the alternation of generations in flowering plants.Biol. Rev.69: 419–442.
Drews, G.N., D. Lee and C.A. Christensen. 1998 Genetic analysis of female gametophyte development and function.Plant Cell10: 5–17.
Fobert, P.R., B.L. Miki and V.N. Iyer, 1981 Detection of gene regulatory signals in plants revealed by T-DNA-mediated fusions.Plant Mal. Biol.17. 837–851.
Greyson, R.I. 1994. The Development of Flowers. New York: Oxford University Press.
Grossniklaus, U., H.J. Bellen, C. Wilson and W.J. Gehring. 1989. P-element mediated enhnacer detection applied to the study of oogenesis inDrosophila.Development107: 189–200.
Grossniklaus, U., R.K. Pearson and W.J. Gehring. 1992. TheDrosophila sloppx paired locus encodes two proteins involved in segmentation that show homology to mammalian transcription factors.Genes and Dev.6: 1030–1051.
Grossniklaus, F. and K. Schneitz. 1998a. The molecular and genetic basis of ovule and megagametopln ie development.Sem in Cell and Dev. Biol.9: 227–238.
Grossniklaus, U., J.M. Moore and W. Gagliano. 1998b. Molecular and genetic approaches to understanding and egineering apomixis: Arabidopsis as a powerful tool. In Hybrid Rice, B. Hardy (ed.) IRRI. Manila. Philippines (in press).
Grossniklaus, U., J-Ph., Vielle-Calzada, M.A. Hoeppner and W.B. Gagliano. 1998c. Maternal control of em-bryogenesis byMEDEA. aPolycomb-group gene inArabidopsis. Science280: 446–450.
Gustafsson, A. 1947. Apomixis in angiosperms II. Funds Univ. Arsskr NF II43: 71–179.
Hermsen, J.G.T. 1984. Mechanisms and genetic implications of 2n-gamele formation lowa StateJ. of Res.58: 421–434.
Huang, B.Q. and S.D. Russell. 1992. The female germ unit. In Sexual reproduction in flowering plants. S.D. Russell and C. Dumas (guest eds.).Int. Rev. Cytol.140: 233–293.
Huang, B.Q. and W.F. Sheridan. 1996. Fmbryo sac development in the maizeindeterminate gametophytel mutant: abnormal nuclear behavior and defective microtubule organization.Plant Cell8: 1391–1407.
Jackson, R.C. and J. Casey. 1980. Cytogenetics of polyploids. In Polyploidy, biological relevance. W.H. Lewis (ed.) Plenum Press. New York pp. 17–44.
Jurgens, G., M. Grebe and T. Steinman. 1997. Establishment of cell polarity during early plant development.Curr. Opin. Cell. Biol.9: 849–852.
Kaul, M.L.H. and T.G.K. Murthy. 1985. Mutant genes affecting higher plant meiosis.Theor. Appl. Genet.70: 449–466.
Kertbundit, S., H. de Greve, F. Deboeck, M. van Montagu and J.-P. Hernalsteens. 1991.ln-vivo random β-glucuronidase gene fusions inArabidopsis thaliana.Proc. Nat. Acad. Sci. USA88: 5212–5216.
Kimber, G. and H. Riley. 1963. Haploid angiosperms.Bot. Rev.29: 480–531
Knox, R.B. 1967. Apomixis: seasonal and population differences in a grass.Science157: 325–326.
Koduru, P.R.K. and M.K. Rao. 1981. Cytogenetics of synaptic mutants.Theor, Appl. Cenet.59: 197–214.
Kojima, A. and Y. Nagato. 1997. Discovery of highly apomictic and highly amphimictic dihaploids inAllium tuberosum.Sex. Pl. Reprod.10: 8–12.
Koltunow, A.M. 1993 Apomixis: embryo sacs and embryos lormed without meiosis or fertilization in ovules.Plant Cell5: 1425–1437.
Koltunow, A.M., K. Soltys, N. Nito and S. McClure. 1995. Anther, ovule, seed and micellar embryo development inCitrus sinensis cv. Valencia.Can. J. Bat.73: 1567–1582.
Ltblanc, O., M.D. Peel, J.G. Carman and Y. Savidan. 1995 Megasporogenesis and megagametogenesis in severalTripsacum species (Poaceae)Am. J. Hot.82: 57–63.
Liu, Y.-G., N. Misukawa, T. Oosumi and R.F. Whittier. 1995. Effcient isolation and mapping ofArabidopsis thaliana T-DNA insert junctions by thermal asym-mertric interlaced PCR.Plant J.8: 457–463.
Maizonnier, D. 1976. Production de tetraploides et de tri-somiques naturels chez le Petunia.Annates d’Amel. des Piantes26: 305–318.
Mascarenhas, J.P. 1992. Pollen gene expression: molecular evidence Pages 3–18 m Sexual Reproduction in flowering Plants. S.D. Russell and C. Dumas eds. International Review of Cvtologv. Vol. 140. Academic Press. New York.
McCormick, S. 1993. Male gametophyte development.Plant Cell5: 1265–1275.
Mogensen, H.L. 1982. Double fertilization in barley and the evtological explanation lor haploid embryo formation. embrvoless caryopses. and ovule abortion.Carlsberg Res. Comm.47: 313–354.
Mogensen, H.L. 1988. Exclusion of male mitochondria and plastids during svngamy in barley as a basis for maternal inheritance.Proc Narl. Acad. Sci. USA85: 2594–2597.
Moore, J.M., J-Ph. Vielle-Calzada, W. Gagliano and U. Grossniklaus. 1997. Genetic characterization of hadad. a mutant disrupting female gametogenesis inArabidopsis tlialiana.Cold Spring Harbor Symp. Quam. Biol.62: 35–47.
Nadeau, J.A., X.S. Zhang, J. Li and S.D. O’Neill. 1996. Ovule development: identification of stage specific and tissue specific cDNAs.Plant Cell8: 213–239.
Naumova T.N. and M.T.M. Willemse. 1995 Ultra-structural characterization of apospory inPanicum maximum.Sex. Plant Reprod.8: 197–204.
Naumova T.N. and J-Ph. Vielle-Calzada. 1998. Ultr-structural analysis of apomictic development. In Apo-mixis: Techniques and Trends. Y. Savidan and J. Carman (eds.). FAO Publications (in press).
Nogler, G.A. 1984a. Gamctophytic apomixis. InEmbryology of angiosperms B.M. Johri (ed.). Springer Verlag. New York, pp. 475–518.
Nogler, G.A. 1984b. Genetics of apospory in apomicticRanvnculus auricomus. V. Conclusion.Bot. Helvet.94: 411–422.
Ohad N., L. Margossian, Y-C. Hsyu, C. Williams, P. Repetti and R.L. Fischer. 1996. A mutation that allows endosperm development without fertilization.Proc. Natl. Acad. Sci. USA93: 5319–5324.
O’Kane, C.J. and W.J. Gehring. 1987. Detection in situ of genomic regulatory elements inDrosophila.Proc. Natl. Acad. Sci. USA85: 9123–9127.
Parrott, W.A. and R.R. Smith. 1986. Production of 2n pollen in red clover.Crop Sci.24: 469–472.
Peel, M.D., J.G. Carman and O. Leblanc. 1997. Megasporocyte callose in apomictic buffelgrass, Kentucky bluegrass,Pennisetum squamulatum Fresen, Tripsacum L.. and weeping lovegrass.Crop Sci.37: 717–723.
Pruitt, R.E., M. Hulskamp, D. Kopczak and K. Schneitz 1994. Genetic analysis of reproductive interactions inArabidopsis. In Pollen-Pistil Interactions and Pollen Tube Growth. A.G. Stephenson and T.H. Kao (eds.)Proc. Ninth Annual Perm State Syrup. Plant Physiol.12: 94–102.
Porter, E.K., D. Parry, J. Bird and H.G. Dickinson. 1984. Nucleic acid metabolism in the nucleus and cytoplasm of angiosperm meiocytes. In Controlling Events in Meiosis. C. Evans and H.G. Dickinson (eds.) Company of Biologists, Cambridge, pp. 363–369.
Ramanna, M.S. 1992. The use of 2n-gametes in breeding polysomic polyploid species; some achievements and perspectives. In Gametes with somatic chromosome number in the evolution and breeding of polyploid polysomic species: achievements and perspectives. A. Mariani and S. Tavoletti (eds.) Perugia, Italy, pp. 91–99.
Rhoades, M.M. and E. Dempsey. 1966. Induction of chromosome doubling by the elongate gene in maize.Genetics54: 505–522.
Russell S.D. 1985. Preferential fertilization inPlumbago: Ultrastructural evidence for gamete-level recognition in an angiosperm.Proc Natl. Acad. Sci. USA82: 6129–6132.
Savidan, Y.H. 1980. Chromosomal and embryological analyses in sexual and apomicitc hybrids ofPanicum maximum Jacq.Theor. Appl. Genet.57: 153–156.
Savidan Y.H. 1992. Progress in research on apomixis and its transfer to major grain crops. InReproductive Biology and Plant Breeding. Y. Dattée and C. Dumas (eds.). Springer-Verlag. Berlin.
Sherwood, R.T., C.C. Berg and B.A. Young. 1994 Inheritance of apospory in buffelgrass.Crop Sci.34: 1490–1494.
Skarnes, W.C. 1990. Entrapment vectors: a new tool for mamalian genetics.Biotechnology8: 827–831.
Smith, D.L., andN.V. Fedoroff. 1995. LRP1. a gene expressed in lateral and adventitious root primordia ofArabidopsis.Plant Cell7, 735–745.
Springer, P.S., W.R. McCombie, V. Sundaresan and R. A. Martienssen. 1995. Gene trap tagging ofprolifera. an essential MCM2-3-5-like gene inArabidopsis.Science268: 877–880.
Sundaresan, V., P. Springer, T. Volpe, S. Haward, J.D. G. Jones, C. Dean, H. Ma and R.A. Martienssen. 1995. Patterns of gene action in plant development revealed by enhancer trap and gene trap transposable elements.Genes & Dcv.9: 1797–1810.
Toppingz, J.F., W. Wei and K. Lindsey, 1991 Func tional tagging of regulatory elements in the plant genome.Development112, 1009–1019.
Twell, D. 1994. The diversity and regulation of gene expression in the pathway of male gametophyte development. In Molecular and Cellular Aspects of Plant reproduction. R. I Scott and A.D. Stead (eds.). Cambridge University Press, Cambridge, pp. 83–136.
Twell D. and R. Howden 1998. Mechanisms of assymetric division and cell fate detrmination in developing pollen. In Haploidy: A critical review. M. Caboche (ed.) Springer Verlag. Berlin, Germany (in press). Veilleux, R. 1985. Diploid and polyploid gametes in crop plants: mechanisms of formation and utilization in plant breeding.Plant Breed. Rev.3: 252–288.
Veronesi, F., A. Mariani and E.T. Bingham. 1986. Unreduced gametes in diploidMedicago and their importance in alfalfa breeding.Theor. Appl. Genet.72: 37–41.
Vielle-Calzada, J-Ph., B.L. Burson, E.C. Bashaw and M.A Hussey. 1995. Early fertilization events in the sexual and aposporous egg apparatus ofPennisetum eiliare (L.) Link.Plant J.8: 309–316.
Vielle-Calzada, J-Ph., C.F. Crane and D.M. Stelly. 1996a. Apomixis: the asexual revolution.Science274: 1322–1323.
Vielle-Calzada, J-Ph., M. Nuccio, M.A. Budiman, T.L. Thomas, B.L. Burson, M.A. Hussey and R.A. Wing. 1996b. Comparative gene expression in sexual and apomictic ovaries ofPennisetum eiliare.Plant Mol. Biol.32: 1085–1092.
Vollbrecht, E. and S. Hake. 1995. Deficiency analysis of female gametogenesis in maize.Dev. Genet.16: 44–63.
Watanabe, K. and S.J. Peloquin. 1989. Occurrence of on pollen and ps gene frequencies in cultivated groups and their related wild species in tuber-bearingSolanums.Theor. Appl. Genet.78: 329–336.
Werner, J.E. and S.J. Peloquin. 1987. Frequency and mechanisms of 2n egg formation in haploid tubero-sum-wild species Fl hybrids.Am. Potato J.64: 641–654.
Willemse, M.T.M. and J.L. Van Went. 1984. The female gametophytc. In Embryology of angiosperms B.M Johri (ed.). Springer Verlag, New York. pp. 159–196.
Wilson, C, R.K. Pearson, H.J. Bellen, C.J. O’Kane, U. Grossniklaus and W.J. Gehring. 1989. P-element mediated enhancer detection: an efficient method for isolating and characterizing developmentally regulated genes.Genes & Dev.3: 1301–1313.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vielle-Calzada, JP., Moore, J.M., Gagliano, W.B. et al. Altering sexual development inArabidopsis . J. Plant Biol. 41, 73–81 (1998). https://doi.org/10.1007/BF03030391
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03030391