Abstract
Purpose
Patient-controlled epidural analgesia (PCEA) with meperidine provides effective analgesia following thoracotomy. Accumulation of normeperidine, a meperidine metabolite with neuroexcitatory effects, has led to recommendations to limit the use of meperidine postoperatively. The purpose of this study was to determine if the addition of adrenaline to PCEA meperidine decreased meperidine consumption, reduced serum normeperidine levels, and improved analgesia following thoracotomy.
Methods
Following Research Ethics approval consenting patients were randomly assigned to PCEA with either meperidine (2 mg·mL-1) + adrenaline (2 μg·mL-1) or meperidine alone (2 mg·mL-1). All patients received a standardized anesthetic and similar perioperative care. Visual analogue pain scores (at rest and with activity), quality of recovery (QoR) scores, and side effects were documented six, 24, and 48 hr postoperatively. Serum levels of meperidine and normeperidine were measured at the same time points.
Results
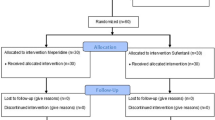
Forty-six patients completed the study protocol. Meperidine consumption (mean ± SD) was similar in the meperidine + adrenaline and the meperidine groups (601 ± 211 mg vs 580 ±211 mg over 48 hr, respectively; P = 0.744). Serum meperidine levels were similar at all study time points. Serum normeperidine was not detected in any sample. Pain scores, QoR scores, and adverse events were comparable in both study groups.
Conclusion
The addition of adrenaline did not influence PCEA meperidine consumption, analgesia outcomes, or QoR. Normeperidine did not accumulate in patients of either study group during the 48-hr study period. Meperidine for patient-controlled epidural analgesia, with or without adrenaline, provides effective post-thoracotomy analgesia in selected patients.
Résumé
Objectif
L’analgésie péridurale contrôlée par le patient (APCP) avec de la mépéridine offre une analgésie efficace après une thoracotomie. L’accumulation de normépéridine, un métabolite de la mépéridine aux effets neuroexcitateurs, a engendré des recommandations dans le but de limiter l’utilisation de mépéridine dans le contexte postopératoire. L’objectifde cette étude était de déterminer si l’adjonction d’adrénaline à la mépéridine APCP réduit la consommation de mépéridine, abaisse les niveaux sériques de normépéridine, et améliore l’analgésie après une thoracotomie.
Méthode
Avec l’assentiment du comité d’éthique de la recherche, les patients consentants ont été randomisés à une APCP avec soit de la mépéridine (2 mg·mL-1) + adrénaline (2 μg·mL-1), soit de la mépéridine seule (2 mg·mL-1). Tous les patients ont reçu un anesthésique standardisé et des soins périopératoires similaires. Les scores de douleur visuels analogues (au repos et à l’effort), les scores de la qualité de la récupération (QoR), et les effets secondaires ont été enregistrés à six, 24 et 48 h postopératoires. Les niveaux sériques de mépéridine et de normépéridine ont été mesurés aux mêmes temps.
Résultats
Quarante-six patients ont terminé le protocole d’étude. La consommation de mépéridine (moyenne ± déviation standard (SD)) était similaire dans les groupes mépéridine + adrénaline et mépéridine seule (601 ± 211 mgvs 580 ±211 mg sur 48 h, respectivement; P = 0,744). Les niveaux sériques de mépéridine étaient similaires à tous les points temporels de l’étude. Aucune normépéridine sérique n’a été détectée dans les échantillons. Les scores de douleur, les scores QoR et les événements indésirables étaient comparables dans les deux groupes à l’étude.
Conclusion
L’ajout d’adrénaline n’a pas influencé la consommation de mépéridine APCP, l’analgésie, ou la QoR. La normépéridine ne s’est accumulée chez les patients déaucun des deux groupes durant la période d’étude de 48 h. La mépéridine pour l’analgésie péridurale contrôlée par le patient, avec ou sans adrénaline, offre une analgésie efficace après une thoracotomie chez certains patients.
Article PDF
Similar content being viewed by others
References
Harpole DHJr,De Camp MM Jr,Daley J, et al. Prognostic models of thirty-day mortality and morbidity after major pulmonary resection. J Thorac Cardiovasc Surg 1999; 117:969–79.
Ballantyne JC, Carr DB, deFerranti S, et al. The comparative effects of postoperative analgesic therapies on pulmonary outcome: cumulative meta-analyses of randomized, controlled trials. Anesth Analg 1998; 86:598–612.
Badner NH, Komar WE. Bupivacaine 0.1% does not improve post-operative epidural fentanyl analgesia after abdominal or thoracic surgery. Can J Anaesth 1992; 39:330–6.
Etches RC, Gammer TL, Cornish R. Patient-controlled epidural analgesia after thoracotomy: a comparison of meperidine with and without bupivacaine. Anesth Analg 1996; 83:81–6.
Ngan Kee WD. Epidural pethidine: pharmacology and clinical experience. Anaesth Intensive Care 1998; 26:247–55.
Jaffe RA, Rowe MA. A comparison of the local anesthetic effects of meperidine, fentanyl, and sufentanil on dorsal root axons. Anesth Analg 1996; 83:776–81.
Ebert B, Andersen S, Krogsgaard-Larsen P. Ketobemidone, methadone and pethidine are non-competitive N-methyl-D-aspartate (NMDA) antagonists in the rat cortex and spinal cord. Neurosci Lett 1995; 187:165–8.
Smith AJ, Haynes TK, Roberts DE, Harmer M. A comparison of opioid solutions for patient-controlled epidural analgesia. Anaesthesia 1996; 51:1013–7.
National Pharmaceutical Council, Joint Commission on Accreditation of Healthcare Organizations. Pain: current understanding of assessment, management, and treatments, 2001. Available from URL; http://www. npcnow.org/resources/PDFs/PainExecSummary.pdf.
Niemi G, Breivik H. Adrenaline markedly improves thoracic epidural analgesia produced by a low-dose infusion of bupivacaine, fentanyl and adrenaline after major surgery. A randomised, double-blind, cross-over study with and without adrenaline. Acta Anaesthesiol Scand 1998; 42:897–909.
BurmAG, van Klee fJW, Gladines MP, Olthof G, Spierdijk J. Epidural anesthesia with lidocaine and bupivacaine: effects of epinephrine on the plasma concentration profiles. Anesth Analg 1986; 65:1281–4.
Baron CM, Kowalski SE, Greengrass R, Horan TA, Unruh HW, Baron CL. Epinephrine decreases postoperative requirements for continuous thoracic epidural fentanyl infusions. Anesth Analg 1996; 82:760–5.
Myles PS, Hunt JO, Nightingale CE, et al. Development and psychometric testing of a quality of recovery score after general anesthesia and surgery in adults. Anesth Analg 1999; 88:83–90.
Ishii A, Tanaka M, Kurihara R, et al. Sensitive determination of pethidine in body fluids by gas chroma -tography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 792:117–21.
Neal JM. Effects of epinephrine in local anesthetics on the central and peripheral nervous systems: neuro-toxicity and neural blood flow. Reg Anesth Pain Med 2003; 28:124–34.
Curatolo M, Petersen-Felix S, Arendt-Nielsen L, Zbinden AM. Epidural epinephrine and clonidine: segmental analgesia and effects on different pain modalities. Anesthesiology 1997; 87:785–94.
Niemi G, Breivik H. The minimally effective concentration of adrenaline in a low-concentration thoracic epidural analgesic infusion of bupivacaine, fentanyl and adrenaline after major surgery. A randomized, double-blind, dose-finding study. Acta Anaesthesiol Scand 2003; 47:439–50.
Niemi G, Breivik H. Epinephrine markedly improves thoracic epidural analgesia produced by a small-dose infusion of ropivacaine, fentanyl, and epinephrine after major thoracic or abdominal surgery: a randomized, double-blinded crossover study with and without epinephrine. Anesth Analg 2002; 94:1598–605.
Bernards CM, Shen DD, Sterling ES, et al. Epidural, cerebrospinal fluid, and plasma pharmacokinetics of epidural opioids (part 2): effect of epinephrine. Anesthesiology 2003; 99:466–75.
Fukuda K. Intravenous opioid anesthetics.In: Miller DR (Ed.). Miller’s Anesthesia 6 ed. Philadelphia: Churchill Livingstone; 2005.
Stone PA, Macintyre PE, Jarvis DA. Norpethidine toxicity and patient controlled analgesia. Br J Anaesth 1993; 71:738–40.
Kussman BD, Sethna NF. Pethidine-associated seizure in a healthy adolescent receiving pethidine for postope-rative pain control. Paediatr Anaesth 1998; 8:349–52.
Mauro VF, Bonfiglio MF, Spunt AL. Meperidine-induced seizure in a patient without renal dysfunction or sickle cell anemia. Clin Pharm 1986; 5:837–9.
Slinger P, Shennib H, Wilson S. Postthoracotomy pulmonary function: a comparison of epidural versus intravenous meperidine infusions. J Cardiothorac Vasc Anesth 1995; 9:128–34.
Ngan Kee WD, Khaw KS, Ma ML. The effect of the addition of adrenaline to pethidine for patient-controlled epidural analgesia after caesarean section. Anaesthesia 1998; 53:1012–6.
Myles PS, Reeves MD, Anderson H, Weeks AM. Measurement of quality of recovery in 5672 patients after anaesthesia and surgery. Anaesth Intensive Care 2000; 28:276–80.
Auquier P, Pernoud N, Bruder N, et al. Development and validation of a perioperative satisfaction questionnaire. Anesthesiology 2005; 102:1116–23.
Heidegger T, Husemann Y, Nuebling M, et al. Patient satisfaction with anaesthesia care: development of a psychometric questionnaire and benchmarking among six hospitals in Switzerland and Austria. Br J Anaesth 2002; 89:863–72.
Author information
Authors and Affiliations
Corresponding author
Additional information
Funding: This research was supported by the 2004 Baxter Corporation Canadian Research Award in Anesthesia, the Ontario Thoracic Society, and the Department of Anesthesiology at the University of Ottawa. Investigators were supported by the Ottawa Hospital Department of Anesthesiology.
Rights and permissions
About this article
Cite this article
Bryson, G.L., Thompson, C., Gagne, S. et al. The addition of adrenaline to thoracic epidural meperidine does not improve analgesia following thoracotomy. Can J Anesth 54, 882–890 (2007). https://doi.org/10.1007/BF03026791
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03026791