Abstract
Purpose
Alpha-adrenergic receptor (AR) agonist drugs (e.g., epinephrine) are commonly used for upper airway procedures, to shrink the mucosa, retard absorption of local anesthetic agents, and improve visualization by limiting hemorrhage. Decongestant therapy often also includes αAR agonist agents, however overuse of these drugs (e.g., oxymetazoline) can result in chronic rhinitis and rebound increases in nasal secretion. Since current decongestants stimulate αARs non-selectively, characterization of αAR subtype distribution in human airway (nasal turbinate) offers an opportunity to refine therapeutic targets while minimizing side-effects. We, therefore, investigated αAR subtype expression in human nasal turbinate within epithelial, duct, gland, and vessel cells using in situ hybridization.
Methods
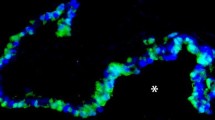
Since sensitive and specific anti-receptor antibodies and highly selective αAR subtype ligands are currently unavailable, in situ hybridization was performed on sections of three human nasal turbinate samples to identify distribution of αAR subtype mRNA. Subtype specific35S-labelled mRNA probes were incubated with nasal turbinate sections, and protected fragments remaining after RNase treatment analyzed by light and darkfield microscopy.
Results
In non-vascular tissue αld AR mRNA predominates, whereas notably the α2c is the only αAR subtype present in the sinusoids and arteriovenous anastamoses.
Conclusion
Combined with the current understanding that AR-mediated constriction of nasal sinusoids underpins decongestant therapies that minimize secretions and shrink tissues for airway procedures, these findings suggest that α2c AR subtypes provide a novel selective target for decongestant therapy in humans.
Objectif
Les médicaments agonistes des récepteurs alphaadrénergiques (AR) (par ex., l’épinéphrine) sont communément utilisés lors des interventions sur les voies aériennes supérieures, afin de rétrécir la muqueuse, de retarder l’absorption d’agents anesthésiques locaux et d’améliorer la visualisation en limitant l’hémorragie. Un traitement décongestionnant inclut également souvent des agents agonistes αAR; toutefois, la surutilisation de ces médicaments (par ex., l’oxymétazoline) peut engendrer une rhinite chronique et l’augmentation rebond des sécrétions nasales lors de la cessation du traitement. Puisque les décongestionnants actuels stimulent les αAR de manière non-sélective, la caractérisation de la distribution des sous-types d’αAR dans les voies aériennes de l’homme (cornet nasal) offre la possibilité de perfectionner les cibles thérapeutiques tout en minimisant les effets secondaires. C’est pourquoi nous avons examiné l’expression des sous-types d’αAR au niveau du cornet nasal humain dans les cellules épithéliales, du canal, des glandes et des vaisseaux, à l’aide d’une hybridation in situ.
Méthode
étant donné que des anticorps anti-récepteurs sensibles et spécifiques ainsi que des ligands très sélectifs des sous-types d’αAR sont disponibles actuellement, l’hybridation in situ a été effectuée sur des sections de trois échantillons de cornet nasal humain afin d’identifier la distribution d’ARN messager des sous-types d’αAR. Des sondes d’ARN messager marquées au35S et spécifiques au sous-type ont été incubées avec des sections de cornet nasal, et les fragments protégés restants après le traitement à la ribonucléase ont été analysés par microscopie optique et sur fond noir.
Résultats
Dans les tissus non-vasculaires, l’ARN messager AR αld est prédominant, alors que le α2c est notablement le seul sous-type d’αAR présent dans les sinusoïdes et les anastomoses artérioveineuses.
Conclusion
On considère maintenant que la constriction des sinus nasaux médiée par AR est à la base des thérapies de décongestion qui minimisent les sécrétions et rapetissent les tissus lors des interventions sur les voies aériennes. Ces résultats suggèrent donc que les sous-types d’AR α2c fournissent une nouvelle cible sélective pour les traitements de décongestion chez les humains.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Mullol J, Raphael GD, Lundgren JD, et al. Comparison of human nasal mucosal secretion in vivo and in vitro. J Allergy Clin Immunol 1992; 89:584–92.
Andersson KE, Bende M. Adrenoceptors in the control of human nasal mucosal blood flow. Ann Otol Rhinol Laryngol 1984; 93 (2Pt 1):179–82.
Malm L. Vascular and secretory effect of adrenoceptor agonists and peptides in the nose. Eur J Respir Dis Suppl 1983; 128 (Pt 1):139–42.
Graf P. Rhinitis medicamentosa: a review of causes and treatment. Treat Respir Med 2005; 4:21–9.
Talaat M, Belal A, Aziz T, Mandour M, Maher A. Rhinitis medicamentosa: electron microscopic study. J Laryngol Otol 1981; 95:125–31.
Su XY, Li Wanpo A. The effect of some commercially available antihistamine and decongestant intra-nasal formulations on ciliary beat frequency. J Clin Pharm Ther 1993; 18:219–22.
van Megen YJ, Klaassen AB, Rodrigues de Miranda JF, van Ginneken CA, Wentges BT. Alterations of adrenoceptors in the nasal mucosa of allergic patients in comparison with nonallergic individuals. J Allergy Clin Immunol 1991; 87:530–40.
Van Megen YJ, Van Ratingen CJ, Klaassen AB, Rodrigues de Miranda JF, Van Ginneken CA, Wentges BT. Biochemical and autoradiographic analysis of beta- adrenoceptors in rat nasal mucosa. Eur J Pharmacol 1990; 182:515–25.
Ishibe T, Yamashita T, Kumazawa T, Tanaka C. Adrenergic and cholinergic receptors in human nasal mucosa in cases of nasal allergy. Arch Otorhinolaryngol 1983; 238:167–73.
Price DT, Lefkowitz RJ, Caron MG, Berkowitz D, Schwinn DA. Localization of mRNA for three distinct alpha 1-adrenergic receptor subtypes in human tissues: implications for human alpha-adrenergic physiology. Mol Pharmacol 1994; 45:171–5.
Rudner XL, Berkowitz DE, Booth JV, et al. Subtype specific regulation of human vascular alpha(1)-adrener- gic receptors by vessel bed and age. Circulation 1999; 100:2336–43.
Stafford-Smith M, Schambra UB, Wilson KH, et al. Alpha 2-adrenergic receptors in human spinal cord: specific localized expression of mRNA encoding alpha 2-adrenergic receptor subtypes at four distinct levels. Brain Res Mol Brain Res 1995; 34:109–17.
Stafford-Smith M, Schambra UB, Wilson KH, Page SO, Schwinn DA. Alpha1-adrenergic receptors in human spinal cord: specific localized expression of mRNA encoding alpha1-adrenergic receptor subtypes at four distinct levels. Brain Res Mol Brain Res 1999; 63:254–61.
Price DT, Chari RS, Berkowitz DE, Meyers WC, Schwinn DA. Expression of alpha 1-adrenergic receptor subtype mRNA in rat tissues and human SK-N-MC neuronal cells: implications for alpha 1-adrenergic receptor subtype classification. Mol Pharmacol 1994; 46:221–6.
Corboz MR, Varty LM, Rizzo CA, et al. Pharmacological characterization of alpha 2-adreno- ceptor-mediated responses in pig nasal mucosa. Auton Autacoid Pharmacol 2003; 23:208–19.
Wang M, Lung MA. Adrenergic mechanisms in canine nasal venous systems. Br J Pharmacol 2003; 138:145- 55.
Ichimura K, Jackson RT. Evidence of alpha 2-adre- noceptors in the nasal blood vessels of the dog. Arch Otolaryngol 1984; 110:647–51.
Tanimitsu N, Yajin K, Sasa M, Tsuru H. Alpha(1)- adrenoceptor subtypes and effect of alpha(1A)-adre- noceptor agonist NS-49 on guinea pig nasal mucosa vasculature. Eur J Pharmacol 2000; 387:73–8.
Berridge TL, Roach AG. Characterization of alpha- adrenoceptors in the vasculature of the canine nasal mucosa. Br J Pharmacol 1986; 88:345–54.
Corboz MR, Rivelli MA, Varty L, et al. Pharmacological characterization of postjunctional alpha-adrenocep- tors in human nasal mucosa. Am J Rhinol 2005; 19:495–502.
Ichimura K, Chow MJ. Postjunctional alpha 2-adreno- ceptors in blood vessels of human nasal mucosa. Arch Otorhinolaryngol 1988; 245:127–31.
Cole P, Haight JS, Cooper PW, Kassel EE. A computed tomographic study of nasal mucosa: effects of vasoac- tive substances. J Otolaryngol 1983; 12:58–60.
Kristiansen AB, Heyeraas KJ, Kirkebo A. Increased pressure in venous sinusoids during decongestion of rat nasal mucosa induced by adrenergic agonists. Acta Physiol Scand 1993; 147:151–61.
Bende M, Elner A, Ohlin P. The effect of provoked allergic reaction and histamine on nasal mucosal blood flow in humans. Acta Otolaryngol 1984; 97:99–104.
Graf P, Toll K, Palm J, Hallen H. Effects of sustained- release oral phenylpropanolamine on the nasal mucosa of healthy subjects. Acta Otolaryngol 1999; 119:837- 42.
Rizzo CA, Ruck LM, Corboz MR, et al. Postjunctional alpha(2C)-adrenoceptor contractility in human saphe- nous vein. Eur J Pharmacol 2001; 413:263–9.
Gavin KT, Colgan MP, Moore D, Shanik G, Docherty JR. Alpha 2C-adrenoceptors mediate contractile responses to noradrenaline in the human saphenous vein. Naunyn Schmiedebergs Arch Pharmacol 1997; 355:406–11.
Chotani MA, Mitra S, Su BY, et al. Regulation of alpha(2)-adrenoceptors in human vascular smooth muscle cells. Am J Physiol Heart Circ Physiol 2004; 286:H59–67.
Flavahan NA. Phenylpropanolamine constricts mouse and human blood vessels by preferentially activating alpha2-adrenoceptors. J Pharmacol Exp Ther 2005; 313:432–9.
Johannssen V, Maune S, Werner JA, Rudert H, Ziegler A. Alpha 1-receptors at pre-capillary resistance vessels of the human nasal mucosa. Rhinology 1997; 35:161–5.
Curtis LN, Carson JL. Computer-assisted video measurement of inhibition of ciliary beat frequency of human nasal epithelium in vitro by xylometazoline. J Pharmacol Toxicol Methods 1992; 28:1–7.
Loth S, Bende M. The effect of topical phenylpropanolamine on nasal secretion and nasal airway resistance after histamine challenge in man. Clin Otolaryngol Allied Sci 1985; 10:15–9.
Malm L, McCaffrey TV, Kern EB. Alpha-adrenoceptor- mediated secretion from the anterior nasal glands of the dog. Acta Otolaryngol 1983; 96:149–55.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stafford-Smith, M., Wilson, K., Bartz, R. et al. Alpha-adrenergic mRNA subtype expression in the human nasal turbinate. Can J Anesth 54, 549–555 (2007). https://doi.org/10.1007/BF03022319
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03022319