Abstract
Purpose
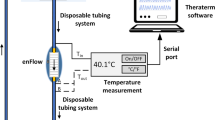
To determine the effect of infusion rate, tubing length and fluid composition on the temperature of the infusate reaching the distal end of an infusion tubing with and without active fluid warming.
Methods
Warmed normal saline (W-NS) and packed red blood cells (W-PRBC), were infused with a fluid warmer through a modified infusion set. The fluids were delivered at eight infusion rates from 50 to 999 mL·hr−1. The infusate temperature was monitored at 20 cm intervals on theiv tubing. The same temperature monitoring protocol was applied to PRBC without warmer (NoW-PRBC).
Results
In W-NS and W-PRBC groups, there was a decrease in the infusate temperature, at each flow rate, from the drip chamber to the distal end of tubing (P < 0.001). In NoW-PRBC group, there was a rapid increase in the infusate temperature from the bag to the drip chamber (P < 0.001). Thereafter, there was no change in temperature, except at the 999 mL·hr−1 infusion rate, where a slight increase in the infusate temperature throughout the tubing was shown. In W-NS and W-PRBC groups increasing the flow rate produced a significant increase in the infusate temperature, at each measurement point (P < 0.001). In the NoW-PRBC group, increasing the flow rate did not alter the infusate temperature. The fluid composition did not influence the infusate temperature.
Conclusion
There is an important heat exchange within the tubing, which is aggravated at low flow rates. At infusion rates appropriate for pediatric anesthesia the clinical and economic value of fluid warming without the use of heated extension tubing is questionable.
Résumé
Objectif
Déterminer l’effet de la vitesse de perfusion, de la longueur de la tubulure et de la composition du liquide sur la température de la solution intraveineuse à la partie distale de la tubulure, le liquide étant soumis ou non à un réchauffement actif.
Méthode
Une solution salée (SS-R) et un concentré de globules rouges réchauffés (CGR-R) ont été perfusés avec un ensemble à perfusion modifié comprenant un réchauffeur de liquide. Les liquides ont été administrés selon huit vitesses de perfusion, de 50 à 999 mL·hr−1. La température du soluté a été mesurée à des intervalles de 20 cm sur la tubulure iv. Le même protocole de surveillance de la température a été appliqué au CGR non réchauffé (CGR-NonR).
Résultats
Dans les groupes SS-R et CGR-R, la température du liquide perfusé a baissé pour toutes les vitesses de perfusion, entre la chambre compte-gouttes et la partie distale de la tubulure (P < 0,001). Dans le groupe CGR-NonR, elle a augmenté rapidement, entre le sac et la chambre compte-gouttes (P < 0,001). Par la suite, il n’y a pas eu de changement de température, sauf à 999 mL·h−1, où on a noté une légère hausse au travers de la tubulure. Dans les groupes SS-R et CGR-R, l’augmentation de la vitesse de perfusion a produit une augmentation significative de la température de la solution intraveineuse, à chaque point de mesure (P < 0,001). Dans le groupe CGR-NonR, l’augmentation de la vitesse d’écoulement n’a pas modifié la température du liquide. La composition de la solution n’a pas changé la température.
Conclusion
Il se produit un important échange de chaleur à l’intérieur de la tubulure, échange qui est augmenté aux basses vitesses de perfusion. à des vitesses de perfusion appropriées à l’anesthésie pédiatrique, la valeur clinique et économique du réchauffement du liquide sans l’utilisation d’une extension chauffante est discutable.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bissonnette B, Davies PJ. Thermal regulation-physiology and perioperative management in infants and children.In: Motoyama ET, Davies PJ (Eds.). Smith’s Anesthesia for Infants and Children. St Louis: CV Mosby; 1995: 139–58.
Huckabee HCG, Craig PL, Williams JM. Near drowning in frigid water: a case study of a 31-year-old woman. J Int Neuropsychol Soc 1996; 2: 256–60.
Schmidt U, Fritz K-W, Kasperczyk W, Tscherne H Successful rescucitation of a child with severe hypothermia after cardiac arrest of 88 minutes. Prehospital Disaster Med 1995; 10: 60–2.
Sessler DI. Mild perioperative hypothermia. N Engl J Med 1997; 336: 1730–7.
Lenhardt R, Marker E, Goll V, et al. Mild intraoperative hypothermia prolongs postanesthetic recovery. Anesthesiology 1997; 87: 1318–23.
Buggy DJ, Crossley AWA. Thermoregulation, mild perioperative hypothermia and postanaesthetic shivering. Br J Anaesth 2000; 84: 615–28.
Bernabei AF, Levison MA, Bender JS. The effects of hypothermia and injury severity on blood loss during trauma laparotomy. J Trauma 1992; 33: 835–9.
Schmied H, Kurz A, Sessler DI, Kozek S, Reiter A. Mild hypothermia increases blood loss and transfusion requirements during total hip arthroplasty. Lancet 1996; 347: 289–92.
Kurz A, Sessler DI, Lenhardt R, for the study of Wound Infection and Temperature Group. Perioperative normothermia to reduce the incidence of surgical-wound infection and shorten hospitalization. Study of wound infection and temperature group. N Engl J Med 1996; 334: 1263–4.
Harrison MJ, Healy TEJ. A comparison of four blood warmers. Anaesthesia 1975; 30: 651–5.
Flancbaum L, Trooskin SZ, Pedersen H. Evaluation of blood-warming devices with the apparent thermal clearance. Ann Emerg Med 1989; 18: 355–9.
Faries G, Johnston C, Pruitt KM, Plouff RT. Temperature relationship to distance and flow rate of warmed iv fluids. Ann Emerg Med 1991; 20: 1198–200.
Schultz J-AI, Sims C, Bissonnette B. Methods for warming intravenous fluid in small volumes. Can J Anaesth 1998; 45: 1110–5.
Presson RG Jr, Bezruczko AP, Hillier SC, McNiece WL. Evaluation of a new fluid warmer effective at low to moderate flow rates. Anesthesiology 1993; 78: 974–80.
Henker R, Bernardo LM, O’Connor K, Sereika S. Evaluation of four methods of warming intravenous fluids. J Emerg Nurs 1995; 21: 385–90.
Norman EA, Ahmad I, Zeig NJ. Delivery temperature of heated and cooled intravenous solutions. Anesth Analg 1986; 65: 693–9.
Kreith F, Bohn MS. Principles of Heat Transfer, 5th ed. New York: West Publishing Co., 1993.
Werner J. Biophysics of heat exchange between body and environment.In: Blatteis C (Ed.). Physiology and Pathophysiology of Temperature Regulation. New Jersey: World Scientific, 1998: 25–45.
Dunham CM, Belzberg H, Lyles R, et al. The rapid infusion system: a superior method for the resuscitation of hypovolemic trauma patients. Resuscitation 1991; 21: 207–27.
Browne DA, de Boeck R, Morgan M. An evaluation of the Level 1 blood warmer series. Anaesthesia 1990; 45: 960–3.
Malcom-Thomas B, Rolly G. Experimental assessment of the Fenwal blood warming system. Acta Anaesthesiol Belg 1974; 1: 100–10.
Presson RG Jr, Haselby KA, Bezruczko AP, Barnett E. Evaluation of a new high-efficiency blood warmer for children. Anesthesiology 1990; 73: 173–6.
Uhl L, Pacini D, Kruskall MS. A comparative study of blood warmer performance. Anesthesiology 1992;77: 1022–8.
Russell WJ. A review of blood warmers for massive transfusion. Anaesth Intensive Care 1974; 2: 109–30.
Manners JM, Mills KLM. Another blood warmer. Some observations of blood changes using the “Hemokinetitherm”. Anaesthesia 1968; 23: 646–56.
Courrèges P, Sorba F, Lecoutre D, Bayart R. Evaluation of the Hotline™ fluid warmer with children under 10 kg (French). Ann Fr Anesth Réanim 1994; 13: 904–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bissonnette, B., Paut, O. Active warming of saline or blood is ineffective when standard infusion tubing is used: an experimental study. Can J Anesth 49, 270–275 (2002). https://doi.org/10.1007/BF03020526
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03020526