Abstract
Purpose
To investigate whether motor evoked potentials (MEP) to transcranial electrical stimulation under constant blood propofol concentration are affected by the arousing effect of surgical noxious stimuli.
Methods
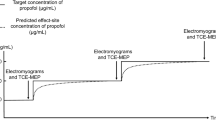
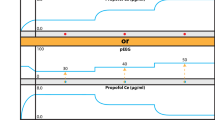
Twenty patients who underwent elective spinal surgery were studied. Patients were anesthetized with 50% nitrous oxide in oxygen, fentanyl, and propofol to maintain the bispectral index (BIS) score around 50. MEP in response to a multipulse transcranial electrical stimulation at stimulus sites of C3–C4 were recorded over the right abductor pollicis brevis muscle. Changes of peak-to-peak amplitude and onset latency of MEP, BIS score before and after surgical stimuli were evaluated. Propofol plasma concentration was measured at the same time points.
Results
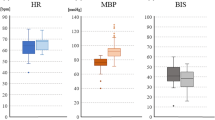
Both MEP amplitude and latency did not change significantly after surgical stimuli although BIS increased significantly (48 ±6 to 58 ± 5;P < 0.05). Plasma propofol concentration was maintained at the same level between the two measurement points (3.3 ± 0.7 to 3.3 ± 0.7 μg·mL−1). There was no relation between BIS change and changes of MEP amplitude and latency, and propofol plasma concentration.
Conclusion
MEP to the transcranial electrical stimulation under a constant and clinically appropriate blood propofol concentration are not affected by surgical noxious stimuli.
Résumé
Objectif
Découvrir si les potentiels évoqués moteurs (PEM) obtenus par une stimulation électrique transcrânienne, pendant le maintien d’une concentration sanguine constante de propofol, sont influencés par l’activation des stimuli chirurgicaux douloureux.
Méthode
Vingt patients qui devaient subir une opération non urgente de la colonne vertébrale ont été étudiés. Ils ont reçu une anesthésie réalisée avec un mélange de protoxyde d’azote et d’oxygène à 50 %, du fentanyl et du propofol pour maintenir la valeur de l’index bispectral (BIS) autour de 50. Les PEM obtenus par une stimulation électrique transcrânienne multi-impulsionnelle aux sites de C3–C4 ont été enregistrés sur le muscle court abducteur du pouce. Les changements d’amplitude crête-à-crête et le temps de latence des PEM ainsi que le score du BIS avant et après les stimuli chirurgicaux ont été évalués. La concentration plasmatique de propofol a été mesurée aux mêmes moments.
Résultats
L’amplitude et la latence des PEM n’ont pas changé de façon significative après les stimuli chirurgicaux, même si le BIS a augmenté significativement (48 ± 6 à 58 ± 5;P < 0,05). La concentration plasmatique de propofol a été maintenue au même niveau entre les deux points de mesure (3,3 ± 0,7 à 3,3 ± 0,7 μg·mL-1). Il n’y avait pas de relation entre les changements du BIS et ceux de l’amplitude et de la latence des PEM, et la concentration plasmatique de propofol.
Conclusion
Les PEM obtenus par stimulation électrique transcrânienne, pendant le maintien d’une concentration sanguine de propofol appropriée à la situation clinique, ne sont pas influencés par les stimuli chirurgicaux douloureux.
Article PDF
Similar content being viewed by others
References
Kalkman CJ, Drummond JC, Ribberink AA, Patel PM, Sano T, Bickford RG. Effects of propofol, etomidate, midazolam, and fentanyl on motor evoked responses to transcranial electrical or magnetic stimulation in humans. Anesthesiology 1992; 76: 502–9.
Kalkman CJ, Drummond JC, Ribberink AA. Low concentrations of isoflurane abolish motor evoked responses to transcranial electrical stimulation during nitrous oxide/opioid anesthesia in humans. Anesth Analg 1991; 73: 410–5.
Kawaguchi M, Inoue S, Kakimoto M, et al. The effect of sevoflurane on myogenic motor-evoked potentials induced by single and paired transcranial electrical stimulation of the motor cortex during nitrous oxide/ketamine/fentanyl anesthesia. J Neurosurg Anesthesiol 1998; 10: 131–6.
Zentner J, Albrecht T, Heuser D. Influence of halothane, enflurane, and isoflurane on motor evoked potentials. Neurosurgery 1992; 31: 298–305.
van Dongen EP, ter Beek HT, Aarts LP, et al. The effect of two low-dose propofol infusions on the relationship between six-pulse transcranial electrical stimulation and the evoked lower extremity muscle response. Acta Anaesthesiol Scand 2000; 44: 799–803.
van Dongen EP, ter Beek HT, Schepens MA, et al. The influence of nitrous oxide to supplement fentanyl/lowdose propofol anesthesia on transcranial myogenic motor-evoked potentials during thoracic aortic surgery. J Cardiothorac Vasc Anesth 1999; 13: 30–4.
Kawaguchi M, Sakamoto T, Inoue S, et al. Low dose propofol as a supplement to ketamine-based anesthesia during intraoperative monitoring of motor-evoked potentials. Spine 2000; 25: 974–9.
Ubags LH, Kalkman CJ, Been HD, Porsius M, Drummond JC. The use of ketamine or etomidate to supplement sufentanil/N2O anesthesia does not disrupt monitoring of myogenic transcranial motor evoked responses. J Neurosurg Anesthesiol 1997; 9: 228–33.
Thees C, Scheufler KM, Nadstawek J, et al. Influence of fentanyl, alfentanil, and sufentanil on motor evoked potentials. J Neurosurg Anesthesiol 1999; 11: 112–8.
Zentner J, Thees C, Pechstein U, Scheufler KM, Wurker J, Nadstawek J. Influence of nitrous oxide on motorevoked potentials. Spine 1997; 22: 1002–6.
Vernon JM, Lang E, Sebel PS, Manberg P. Prediction of movement using bispectral electroencephalographic analysis during propofol/alfentanil or isoflurane/alfentanil anesthesia. Anesth Analg 1995; 80: 780–5.
Sebel PS, Lang E, Rampil IJ, et al. A multicenter study of bispectral electroencephalogram analysis for monitoring anesthetic effect. Anesth Analg 1997; 84: 891–9.
Struys MM, De Smet T, Versichelen LF, Van de Velde S, Van den Broecke R, Mortier EP. Comparison of closedloop controlled administration of propofol using bispectral index as the controlled variable versus “standard practice” controlled administration. Anesthesiology 2001; 95: 6–17.
Kalkman CJ, Drummond JC, Kennelly NA, Patel PM, Partridge BL. Intraoperative monitoring of tibialis anterior muscle motor evoked responses to transcranial electrical stimulation during partial neuromuscular blockade. Anesth Analg 1992; 75: 584–9.
Rampil IJ, Holzer JA, Quest DO, Rosenbaum SH, Correll JW. Prognostic value of computerized EEG analysis during carotid endarterectomy. Anesth Analg 1983; 62: 186–92.
Zhou HH, Kelly PJ. Transcranial electrical motor evoked potential monitoring for brain tumor resection. Neurosurgery 2001; 48: 1075–81.
van Dongen EP, ter Beek HT, Schepens MA, et al. Within patient variability of lower extremity muscle responses to transcranial electrical stimulation with pulse trains in aortic surgery. Clin Neurophysiol 1999; 110: 1144–8.
Woodforth IJ, Hicks RG, Crawford MR, Stephen JP, Burke DJ. Variability of motor-evoked potentials recorded during nitrous oxide anesthesia from the tibialis anterior muscle after transcranial electrical stimulation. Anesth Analg 1996; 82: 744–9.
Antognini JF, Schwartz K. Exaggerated anesthetic requirements in the preferentially anesthetized brain. Anesthesiology 1993; 79: 1244–9.
Rampil IJ, Mason P, Singh H. Anesthetic potency (MAC) is independent of forebrain structures in the rat. Anesthesiology 1993; 78: 707–12.
Antognini JF, Carstens E, Tabo E, Buzin V. Effect of differential delivery of isoflurane to head and torso on lumbar dorsal horn activity. Anesthesiology 1998; 88: 1055–61.
Antognini JF, Wang XW, Piercy M, Carstens E. Propofol directly depresses lumbar dorsal horn neuronal responses to noxious stimulation in goats. Can J Anesth 2000; 47: 273–9.
Zhou HH, Jin TT, Qin B, Turndorf H. Suppression of spinal cord motoneuron excitability correlates with surgical immobility during isoflurane anesthesia. Anesthesiology 1998; 88: 955–61.
Antognini JF, Carstens E, Buzin V. Isoflurane depresses motoneuron excitability by a direct spinal action: an Fwave study. Anesth Analg 1999; 88: 681–5.
Casati A, Fanelli G, Casaletti E, et al. Clinical assessment of target-controlled infusion of propofol during monitored anesthesia care. Can J Anesth 1999; 46: 235–9.
Luginbuhl M, Schnider TW. Detection of awareness with the bispectral index: two case reports. Anesthesiology 2002; 96: 241–3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial support: this work was supported by Grant-in Aid for Scientific Research C2-10671439, Ministry of Education, Tokyo, Japan.
Rights and permissions
About this article
Cite this article
Inoue, S., Kawaguchi, M., Takahashi, M. et al. Noxious stimuli do not modify myogenic motor evoked potentials by electrical stimulation during anesthesia with propofol-based anesthesia. Can J Anesth 50, 86–91 (2003). https://doi.org/10.1007/BF03020194
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03020194