Abstract
Purpose: To seek behavioural, reflexive and histochemical evidence of long-lasting changes in nociceptive stimulus transmission induced by exposure to doses of pentobarbital that induce nocifensive hyperreflexia.
Methods: Nocifensive hyperreflexia was induced in 12 rats with 30 mg·kg−1 pentobarbitalip. Reflex latency times for withdrawal of the hind paw from noxious radiant heat were measured with an automated electronic timer. Subjective responses to noxious stimulation (licking or biting of the stimulated hindpaw) and the level of sedation were recorded. Histological sections of lumbar spinal cord were stained for immunoreactivity of the immediate-early-gene (IEG0, c-fos, in three rats that repeated threshold noxious radiant heat stimulation during the period of nocifensive hyperreflexia induced by 30 mg·kg−1 pentobarbitalip.
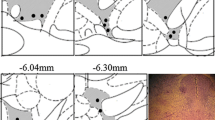
Results: Reflex withdrawal latency decreased by 32±8% of control values (P<0.001) following pentobarbital injection and returned to control values 120 min after drug injection. Once fully alert, pentobarbital-treated animals did not show any increase in nociceptive behaviour relative to saline-injected controls (P=0.41). Sustained noxious stimulation to the hindpaw in halothane-anesthetized animals was associated with an increase inc-fos immunoreactivity in the dorsal horn of the lumbar spinal cord ipsilateral to the stimulation (P<0.001). Threshold stimulation in the pentobarbital-treated animals was not associated with any increase inc-fos expression.
Conclusions: During pentobarbital-induced hyperreflexia, rats did not show any reflexive, behavioural, or histochemical evidence of long-lasting enhancement of nocifensive signal transmission. The results are consistent with previous observations that, in the absence of tissue injury, nocifensive hyperreflexia induced by barbiturates is a short-lived pharmacological effect.
Résumé
Ojectif: Découvrir les manifestations comportementales, réflexes et histochimiques de modifications persistantes de la transmission d’un stimulus nociceptif induit par l’exposition à des doses de pentobarbital qui provoquent une surréflectivité défensive.
Méthode: La surréflectivité défensive a été induite chez 12 rats avec 30 mg·kg−1 de pentobarbitalip. Les temps de latence réflexe nécessaire au retrait de la patte arrière d’une source de chaleur radiante ont été mesurés avec un chronomètre électronique automatisé. Les réponses subjectives à la stimulation désagréable (lécher ou mordre la patte stimulée) et le niveau de sédation ont été enregistrés. Des sections histologiques de la moelle épinière lombaire ont été colorées pour vérifier l’immunoréactivité du gène précoce une stimulation liminale novice répétée de chaleur radiante pendant la période de surréflectivité défensive induite par les 30 mg·kg−1 de pentobarbitalip.
Résultats: Le temps de latence réflexe a baissé de 32±8 % par rapport aux valeurs témoins (P<0,001) après l’injection de pentobarbital et est revenu aux valeurs témoins 120 min après l’injection du médicament. Une fois complètement réveillés, les animaux traités au pentobarbital n’ont pas affiché de comportement nociceptif accru comparés aux animaux témonis à qui on a injecté une solution salée (P=0,41). La stimulation nocive à la patte arrière, subie par les animaux anesthésiés à l’halothane, a été associée avec un accroissement de l’immunoréactivité au gènec-fos dans la corne supérieure de la moelle épinière lombaire homolatérale à la stimulation (P<0,001). La stimulation liminale chez les animaux traités au pentobarbital n’était pas accompagnée d’une augmentation de l7rsexpression dec-fos.
Conclusion: Pendant la surréflectivité liée au pentobarbital, les rats n’ont pas donné de signe réflexe, comportemental ou histochimique d’une augmentation persistante de la transmission du signal défensif. Ces résultats confirment des observations antérieures qui montraient qu’en l’absence de l’sion aux tissus la surréflectivité défensive induite par les barbituriques présente un effet pharmacologique de courte durée.
Article PDF
Similar content being viewed by others
References
Arora MV, Carson IW, Dundee JW. Alterations in response to somatic pain associated with anaesthesia. XXI: Althesin (CT 1341). Br J Anaesth 1972; 44: 590–2.
Briggs LP, Dundee JW, Bahar M, Clarke RSJ. Comparison of the effect of diisopropyl phenol (ICI 35 868) and thiopentone on response to somatic pain. Br J Anesth 1982; 54: 307–11.
Dundee JW. Alterations in response to somatic pain associated with anaesthesia. II: The effect of thiopentone and pentobarbitone. Br J Anaesth 1960; 32: 407–14.
Dundee JW, Moore J. Alterations in response to somatic pain associated with anaesthesia. IV: The effect of subanaesthetic concentrations of inhalation agents. Br J Anaesth 1960; 32: 453–9.
Archer DP, Roth SH. Pharmacodynamics of thiopentone: nocifensive reflex threshold changes correlate with hippocampal electroencephalography. Br J Anaesth 1997; 79: 744–9.
Archer DP, Roth SH. Pentobarbital facilitates long-lasting synaptic potentiation. (Abstract) Society for Neuroscience 1999; 29: 899.3.
Archer DP, Roth SH. Acetazolamide and amiloride inhibit pentobarbital-induced facilitation of nocifensive reflexes. Anesthesiology 1999; 90: 1158–64.
Bullitt E. Expression ofc-fos-like protein as a marker for neuronal activity following noxious stimulation in the rat. J Comp Neurol 1990; 296: 517–30.
Hou w-Y, Shyu B-C, Chen T-M, Lee J-W, Shieh J-Y, Sun W-Z. Intrathecally administeredc-fos antisense oligodeoxynucleotide decreases formalin-induced nociceptive behavior in adult rats. Eur J Pharmacol 1997; 329: 17–26.
Hunt SP, Pini A, Evan G. Induction ofc-fois-like protein in spinal cord neurons following sensory stimulation. Nature 1987; 328: 632–4.
Archer DP, Samanani N, Roth SH. Nocifensive reflex thresholds in rats: measures of central nervous system effects of barbiturates. Can J Anaesth 1997; 44: 765–74.
Hargreaves K, Dubner R, Brown F, Flores C, Joris J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 1988; 32: 77–88.
Ewen A, Archer DP, Samanani N, Roth SH. Hyperalgesia during sedation: effects of barbiturates and propofol in the rat. Can J Anaesth 1995; 42: 532–40.
Goto T, Marota JJA, Crosby G. Pentobarbitone, but not propofol, produces pre-emptive analgesia in the rat formalin model. Br J Anaesth 1994; 72: 662–7.
Woolf CJ, McMahon SB. Injury-induced plasticity of the flexor reflex in chronic decerebrate rats. Neuroscience 1985; 16: 395–404.
Yashpal K, Mason P, McKenna JE, Sharma SK, Henry JL, Coderre TJ. Comparison of the effects of treatment with intrathecal lidocaine given before and after formalin on both nociception and Fos expression in the spinal cord dorsal horn. Anesthesiology 1998; 88: 157–64.
Anker-Møller E, Spangsberg N, Arendt-Nielsen L, Schultz P, Kristensen MS, Bjerring P. Subhypnotic doses of thiopentone and propofol cause analgesia to experimentally induced acute pain. Br J Anaesth 1991; 66: 185–8.
Dubner R, Ruda MA. Activity-dependent neuronal plasticity following tissue injury and inflammation. Trends Neurosci 1992; 15: 96–101.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported by the Medical Research Council of Canada (S.H.R.) and by the Canadian Anaesthetists’ Society — Burroughs-Wellcome Research Award (D.P.A.).
Rights and permissions
About this article
Cite this article
Archer, D.P., Samanani, N. & Roth, S.H. Pentobarbital induces nocifensive hyperrflexia, not hyperalgesia in rats. Can J Anaesth 47, 687–692 (2000). https://doi.org/10.1007/BF03019003
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03019003