Abstract
Purpose
To examine the hypothesis that transthoracic echocardiographic findings predict mortality in critically ill patients.
Methods
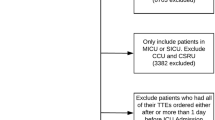
A retrospective analysis of concurrently collected data for consecutive patients from May 1996 to May 1998 who had transthoracic echocardiography on or within six months of admission to the medical surgical intensive care (MSICU). We examined the role of physiologic, clinical, and echocardiography variables in predicting the mortality of patients admitted to the MSICU. Three logistic regression models were developed: 1) clinical; 2) echocardiographic; and 3) combined clinical with echocardiographic. Univariate and multivariate analyses were performed and the relative strength of clinical and echocardiographic predictors was compared using odds ratio (OR) and receiver-operator-characteristic (ROC).
Results
Of 4,070 MSICU patient admissions, 1,093 patients had transthoracic echocardiography; the study group comprised 942 patients with complete clinical and echocardiographic data. The MSICU mortality was 28%. For the combined model, analyses identified left ventricular systolic function (LVSF), OR 1.26; confidence interval (Cl) 1.01–1.57, severe tricuspid regurgitation (TR) (OR 3.72; Cl 1.04–13.24), medical diagnosis (OR 1.91; Cl 1.15–3.19), and acute physiology and chronic health evaluation (APACHE) II score (OR 1.27; Cl 1.23–1.31), as predictors of MSICU mortality. The combined model yielded an area under ROC curve of 0.913. For the clinical model, analyses identified age (OR 1.04; Cl 1.02–1.05) and APACHE II (OR 1.32; 1.26–1.35) as predictors of mortality with an area under ROC curve of 0.917. For the echocardiography model, TR (OR 2.40; 1.08–5.38), severe aortic insufficiency (Al) (OR 4.13; Cl 1.17–16.29) and pulmonary hypertension (OR 2.05; 1.01–4.09) were identified as predictors of outcome with an ROC curve of 0.536 for this model.
Conclusion
Statistical models utilizing clinical variables are predictive of mortality in MSICU. Models that include diagnostic transthoracic echocardiography variables do not provide incremental value to predict ICU mortality. These findings may have implications for non-invasive hemodynamic assessment of critically ill patients, and raise the hypothesis that echocardiography-guided interventions may not alter outcome in ICU.
Résumé
Objectif
Vérifier l’hypothèse selon laquelle les résultats de l’échocardiographie transthoracique permettent de prédire la mortalité chez les grands malades.
Méthode
Une analyse rétrospective a été faite des données recueillies simultanément auprès de patients successifs qui ont eu, entre mai 1996 et mai 1998, une échographie transthoracique six mois ou moins après l’admission à l’unité des soins intensifs médicaux chirurgicaux (USIMC). Nous avons vérifié le rôle des variables physiologiques, cliniques et échocardiographiques dans la prédiction de la mortalité à l’USIMC. Trois modèles de régression logistique ont été élaborés: clinique, échocardiographique, et clinique et échocardiographique combiné. Des analyses à une ou plusieurs variables ont été réalisées et la valeur relative des prédicteurs cliniques et échocardiographiques a été comparée selon le risque relatif (RR) et la courbe ROC.
Résultats
Des 4 070 patients admis à l’USIMC, 1093 ont eu une échocardiographie transthoracique; le groupe expérimental comprenait 942 patients dont nous avions les données cliniques et échocardiographiques complètes. La mortalité à l’USIMC a été de 28 %. Pour le modèle combiné, la fonction systolique du ventricule gauche (FSVG), RR de 1,26, Intervalle de confiance (lC) de 1,01–1,57, la régurgitation tricuspide (RT) sévère (RR de 3,12; lC de 1,04–13,24), le diagnostic médical (RR de 1,91; lC de 1, 15–3,19) et le score APACHE II (RR de 1,27 ; Cl de 1,23–1,31) ont été des prédlcteurs de mortalité à l’USIMC. Ce modèle présentait une aire sous la courbe ROC de 0,913. Pour le modèle clinique, l’âge (RR de 1,04 ; lC de 1,02–1,05) et le score APACHE II (RR de 1,32 ; 1,26–,35) ont été des prédlcteurs de mortalité avec une aire sous la courbe de 0,917. Pour le modèle échocardlographlque, la RT (RR de 2,40; 1,08–5,38), l’insuffisance aortlque sévère (lA) (RR de 4,13; Cl de 1,17–16,29) et l’hypertension pulmonaire (RR de 2,05; 1,01–4,09) ont été des prédlcteurs avec une aire sous la courbe de 0,536.
Conclusion
Les modèles statistiques utilisant des variables cliniques sont prédictifs de mortalité à l’USIMC. Les modèles Incluant les variables diagnostiques de l’échocardlographie transthoraclque n’améliore pas la prédiction de la mortalité à l’USl. Ces résultats peuvent infuencer l’évaluation hémodynamique non effractlve des grands malades et donner à penser que des interventions guidées par échocardlographle ne modifient pas l’évolution à l’USI.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Schuster DP. Predicting outcome after ICU admission. The art and science of assessing risk. Chest 1992; 102: 1861–70.
Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med 1985; 13: 818–29.
Krumholz HM, Douglas PS, Goldman L, Waksmonski C. Clinical utility of transthoracic two-dimensional and Doppler echocardiography. J Am Coll Cardiol 1994; 24: 125–31.
Vignon P, Mentec H, Terre S, Gastinne H, Gueret P, Lemaire F. Diagnostic accuracy and therapeutic impact of transthoracic and transesophageal echocardiography in mechanically ventilated patients in the ICU. Chest 1994; 106: 1829–34.
Kim R, Chakko S, Myerburg RJ, Kessler KM. Clinical usefulness and cost of echocardiography in patients admitted to a coronary care unit. Am J Cardiol 1997; 80: 1273–6.
Xie GY, Berk MR, Smith MD, Gurley JC, DeMaria AN. Prognostic value of Doppler transmitral flow patterns in patients with congestive heart failure. J Am Coll Cardiol 1994; 24: 132–9.
Stamm RB, Carabello BA, Mayers DL, Martin RP. Two-dimensional echocardiographic measurement of left ventricular ejection fraction: prospective analysis of what constitutes an adequate determination. Am Heart J 1982;104: 136–44.
Skjaerpe T, Hegrenaes L, Hatle L. Noninvasive estimation of valve area in patients with aortic stenosis by Doppler ultrasound and two-dimensional echocardiography. Circulation 1985; 72: 810–8.
Hatle L, Angehen B. Pulsed and continuous wave Doppler in diagnosis and assessment of various heart lesions. In: Hatle L, Angelsen B, (Eds.). Doppler Ultrasound in Cardiology: Physical Principles and Clinical Applications, 2nd ed. Philadelphia: Lea and Febiger; 1982.
Miyatake K, Izumi S, Okamoto M, et al. Semiquantitative grading of severity of mitral regurgitation by real-time two-dimensional Doppler flow imaging technique. J Am Coll Cardiol 1986; 7: 82–8.
Perry GJ, Helmcke F, Nanda NC, Byard C, Soto B. Evaluation of aortic insufficiency by Doppler color flow mapping. J Am Coll Cardiol 1987; 9: 952–9.
Currie PJ, Seward JB, Chan KL, et al. Continuous wave Doppler determination of right ventricular pressure: a simultaneous Doppler-catheterization study in 127 patients. J Am Coll Cardiol 1985; 6: 750–6.
Knaus WA, Wagner DP, Draper EA, et al. The APACHE III prognostic system. Risk prediction of hospital mortality for critically ill hospitalized adults. Chest 1991; 100: 1619–36.
Wong DT, Crofts SL, Gomez M, McGuire GP, Byrick RJ. Evaluation of predictive ability of APACHE II system and hospital outcome in Canadian intensive care unit patients. Crit Care Med 1995; 23: 1177–83.
Zimmerman JE, Knaus WA, Judson JA, et al. Patient selection for intensive care: a comparison of New Zealand and United States hospitals. Crit Care Med 1988; 16: 318–26.
Giangiuliani G, Mancini A, Gui D. Validation of a severity of illness score (APACHE II) in a surgical intensive care unit. Intensive Care Med 1989; 15: 519–22.
Sirio CA, Tajimi K, Tase C, et al. An initial comparison of intensive care in Japan and the United States. Crit Care Med 1992; 20: 1207–15.
Oh TE, Hutchinson R, Short S, Buckley T, Lin E, Leung D. Verification of the acute physiology and chronic health evaluation scoring system in a Hong Kong intensive care unit. Crit Care Med 1993; 21: 698–705.
Hwang JJ, Shyu ICG, Chen JJ, Tseng YZ, Kuan P, Lien WP. Usefulness of transesophageal echocardiography in the treatment of critically ill patients. Chest 1993; 104: 861–6.
Slama MA, Novara A, Van De Putte P, et al. Diagnostic and Therapeutic Implications of transesophageal echocardiography in medical ICU patients with unexplained shock, hypoxemia, or suspected endocarditis. Intensive Care Med 1996; 22: 916–22.
Tam JW, Nichol J, MacDiarmid AL, Lazarow N, Wolfe K. What is the real clinical utility of echocardiography? A prospective observational study. J Am Soc Echocardiogr 1999; 12: 689–97.
Nishimura RA, Abel MD, Hatle LV, Tajik AJ. Relation of pulmonary vein to mitral flow velocities by transesophageal Doppler echocardiography. Effect of different loading conditions. Circulation 1990; 81: 1488–97.
Kusumoto FM, Muhiudeen IA, Kuecherer HF, Cahalan MIC, Schiller NB. Response of the interatrial septum to transatrial pressure gradients and its potential for predicting pulmonary capillary wedge pressure: an intraoperative study using transesophageal echocardiography in patients during mechanical ventilation. J Am Coll Cardiol 1993; 21: 721–8.
Cheung AT, Savino JS, Weiss SJ, Aukburg SJ, Berlin JA. Echocardiographic and hemodynamic indexes of left ventricular preload in patients with normal and abnormal ventricular function. Anesthesiology 1994; 81: 376–87.
Harpole DH, Clements FM, Quill T, Wolfe WG, Jones RH, McCann RL. Right and left ventricular performance during and after abdominal aortic aneurysm repair. Ann Surg 1989; 209: 356–62.
Gorscan J III, Diana P, Ball BB, Hattler BG Intraoperative determination of cardiac output by transesophageal continuous wave Doppler. Am Heart J 1992; 123; 171–6.
Stoddard MF, Prince CR, Ammash N, Goad JL, Vogel RL. Pulsed Doppler transesophageal echocardiographic determination of cardiac output in human beings: comparison with pulsed thermodilution technique. Am Heart J 1993; 126: 956–62.
Smith JS, Cahalan MK, Benefiel DJ et al. Intraoperative detection of myocardial ischemia in high-risk patients: electrocardiography versus two-dimensional transesophageal echocardiography. Circulation 1985; 72: 1015–21.
Savage RM, Lytle BW, Aronson S, et al. Intraoperative echocardiography is indicated in high-risk coronary artery bypass grafting. Ann Thorac Surg 1997; 64: 368–74.
Heidenreich PA, Stainbach R, Redberg RF, Schiller NB, Cohen NH, Foster E. Transesophageal echocardiography predicts mortality in critically ill patients with hypotension. J Am Coll Cardiol 1995; 26: 152–8.
Wake PJ, Ali M, Carroll J, Siu SC, Cheng DC. Clinical and echocardiographic diagnoses disagree in patients with unexplained hemodynamic instability after cardiac surgery. Can J Anesth 2001; 48: 778–83.
Connors AF Jr, Speroff T, Dawson NV, et al. The effectiveness of right heart catheterization in the initial care of critically ill patients. SUPPORT Investigators. JAMA 1996; 276: 889–97.
Parker MM, Cunnion RE, Parrillo JE. Echocardiography and nuclear cardiac imaging in the intensive care unit. JAMA 1985; 254: 2935–39.
Poelaert J, Schmidt C, Colardyn F. Transoesophageal echocardiography in the critically ill. Anaesthesia 1998; 53: 55–68.
Shanewise JS, Cheung AT, Aronson S, et al. ASE/SCA guidelines for performing a comprehensive intraoperative multiplane transesophageal echocardiography examination: recommendations of the American Society of Echocardiography Council for Intraoperative Echocardiography and the Society of Cardiovascular Anesthesiologists Task Force for Certification in Perioperative Transesophageal Echocardiography. J Am Soc Echocardiogr 1999; 12: 884–900.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by: The Department of Anesthesia, University Health Network, University of Toronto. Toronto, Ontario, Canada.
Rights and permissions
About this article
Cite this article
Sawchuk, C.W.T., Wong, D.T., Kavanagh, B.P. et al. Transthoracic echocardiography does not improve prediction of outcome over APACHE II in medical-surgical intensive care. Can J Anaesth 50, 305–310 (2003). https://doi.org/10.1007/BF03017803
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03017803