Abstract
Purpose: The need for muscular relaxation to improve intubat-ing conditions and to reduce the incidence of laryngeal morbidity is still controversial. The aim of this study was to determine the incidence of symptomatic laryngeal injuries (SLI) and of acceptable intubating conditions (including both good and excellent conditions), both with and without cisatracurium during induction of anesthesia, along with moderate doses of remifen-tanil and propofol.
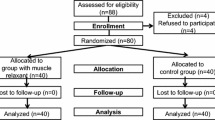
Methods: In this prospective, randomized double-blind equivalence trial, the intubating conditions were compared in 130 ASA I or II female patients. All subjects received remifentanil 2 µg·kg−1iv and propofol 2.5 mg·kg−1iv, with either cisatracu-rium 0.15 mg·kg−1iv (group Cisatracturium), or saline (group Placebo). Tracheal intubating conditions were assessed with the Copenhagen Score. A systematic screening for postoperative hoarseness and sore throat was performed 24 and 48 hr after anesthesia, followed by a nasofibroscopic examination when laryngeal symptoms persisted at 48 hr.
Results: Twenty-four hr after anesthesia, the incidence of postoperative hoarseness and sore throat in the Cisatracurium and Placebo groups was 26.5% and 21.5%, respectively, and 48 hr after anesthesia, the incidence was 7.8% and 6.1%, respectively (P=0.32 andP=0.50 between groups, respectively). In the clinically evaluable population, the incidence of SLI, assessed at 48 hr by nasofibroscopy, was equivalent in both groups, 1.6%vs 1.5% in group Placebo and group Cisatracurium, respectively (P<0.001 for equivalence test), as was the occurrence of acceptable intubating conditions (95.4%vs 100%,P<0.05 for equivalence test). However, the occurrence of excellent intu-bating conditions was more frequent in group Cisatracurium than in group Placebo (P=0.0003).
Conclusion: Following induction of anesthesia with propofol and moderate-dose remifentanil, cisatracurium did not confer a higher rate of good-to-excellent conditions for tracheal intubation, nor did muscle relaxation with cisatracurium decrease the rate of SLI after tracheal intubation.
Résumé
Objectif: Le besoin de relaxation musculaire dans le but d’améliorer les conditions d’intubation et de réduire l’incidence de morbidité laryngée demeure controversé. L’objectif de cette étude était de déterminer l’incidence de lésions laryngées symptomatiques et des conditions d’intubation acceptables (incluant les conditions bonnes et excellentes), avec ou sans cisatracurium pendant l’induction de l’anesthésie, et combiné à des doses modérées de rémifentanil et de propofol.
Méthode: Dans cette étude d’équivalence prospective, randomisée et à double insu, les conditions d’intubation ont été comparées chez 130 patientes ASA I ou II. Toutes les patientes ont reçu 2 µg·kg−1 de rémifentanil iv et 2.5 mg·kg−1 de propofol iv, avec soit 0,15 mg·kg−1 de cisatracurium iv (groupe Cisatracurium), soit une solution saline (groupe Placebo). Les conditions d’intubation trachéale ont été évaluées à l’aide de l’échelle de Copenhague. Un dépistage systématique de l’enrouement et des maux de gorge postopératoires a été réalisé 24 et 48 h après l’anesthésie, suivi d’un examen nasofibros-copique lorsque les symptômes laryngés persistaient à 48 h.
Résultats: L’incidence d’enrouement et de maux de gorge postopératoires à 24 h était de 26,5 % et 21,5 % dans les groupes Cisatracurium et Placebo, respectivement. Quarante-huit heures après l’anesthésie, l’incidence était de 7,8 % et 6,1 %, respectivement (P=0,32 et P=0,50 entre les groupes, respectivement). Dans la population évaluable cliniquement, l’incidence de lésions laryngées symptomatiques, évaluée à 48 h par nasofibrosco-pie, était équivalente dans les deux groupes, soit 1,6 % et 1,5 % dans le groupe Placebo et le groupe Cisatracurium, respectivement (P<0,001 pour le test d’équivalence), tout comme l’occurrence avec des conditions d’intubation acceptable (95,4 % vs 100 %, P<0,05 pour le test d’équivalence). Toutefois, l’occurrence dans les situations où les conditions d’intubation étaient excellentes était plus fréquente dans le groupe Cisatracurium que dans le groupe Placebo (P=0,0003).
Conclusion: À la suite d’une induction de l’anesthésie à l’aide de propofol et de rémifentanil en dose modérée, le cisatracurium n’a pas procuré un taux plus élevé de conditions bonnes à excellentes pour l’intubation trachéale ; en outre, la relaxation musculaire procurée par le cisatracurium n’a pas non plus réduit le taux de lésions laryngées symptomatiques après l’intubation trachéale.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baillard C, Adnet F, Borron SW, et al. Tracheal intubation in routine practice with and without muscular relaxation: an observational study. Eur J Anaesthesiol 2005; 22: 672–7.
Higgins PP, Chung F, Mezei G. Postoperative sore throat after ambulatory surgery. Br J Anaesth 2002; 88: 582–4.
Christensen AM, Willemoes-Larsen H, Lundby L, Jakobsen KB. Postoperative throat complaints after tracheal intubation. Br J Anaesth 1994; 73: 786–7.
Mencke T, Echternach M, Plinkert PK, et al. Does the timing of tracheal intubation based on neuromuscular monitoring decrease laryngeal injury? A randomized, prospective, controlled trial. Anesth Analg 2006; 102: 306–12.
Mencke T, Echternach M, Kleinschmidt S, et al. Laryngeal morbidity and quality of tracheal intubation. A randomized controlled trial. Anesthesiology 2003; 98: 1049–56.
Combes X, Andriamifdy L, Dufresne E, et al. Comparison of two induction regimens using or not using muscle relaxant: impact on postoperative upper airway discomfort. Br J Anaesth 2007; 99: 276–81.
Mertes PM, Laxenaire MC, Alla F;Groupe d’Études des Réactions Anaphylactoïdes Peranesthésiques. Anaphylactic and anaphylactoid reactions occurring during anesthesia in France in 1999-2000. Anesthesiology 2003; 99: 536–45.
Debaene B, Plaud B, Dilly MP, Donati F. Residual paralysis in the PACU after a single intubating dose of nondepolarizing muscle relaxant with an intermediate duration of action. Anesthesiology 2003; 98: 1042–8.
Sandin RH, Enlund G, Samuelsson P, Lennmarken C Awareness during anaesthesia: a prospective case study. Lancet 2000; 355: 707–11.
McNeil IA, Culbert B, Russell I. Comparison of intubating conditions following propofol and succinylcholine with propofol and remifentanil 2 micrograms kg−1 or 4 micrograms kg−1. Br J Anaesth 2000; 85: 623–5.
Stevens JB, Wheatley L. Tracheal intubation in ambulatory surgery patients: using remifentanil and propofol without muscle relaxants. Anesth Analg 1998; 86: 45–9.
Grant S, Noble S, Woods A, Murdoch J, Davidson A. Assessment of intubating conditions in adults after induction with propofol and varying doses of remifentanil. Br J Anaesth 1998; 81: 540–3.
Durmus M, Ender G, Kadir BA, Nurcin G, Erdogan O, Ersoy MO. Remifentanil with thiopental for tracheal intubation without muscle relaxants. Anesth Analg 2003; 96: 1336–9.
Erhan E, Ugur G, Alper I, Gunusen I, Ozyar B. Tracheal intubation without muscle relaxants: remifentanil or alfentanil in combination with propofol. Eur J Anaesthesiol 2003; 20: 37–43.
Alexander R, Booth J, Olufolabi AJ, El-Moalem HE, Glass PS. Comparison of remifentanil with alfentanil or suxamethonium following propofol anaesthesia for tracheal intubation. Anaesthesia 1999; 54: 1032–6.
Scheller MS, Zornow MH, Saidman LJ. Tracheal intubation without the use of muscle relaxants: a technique using propofol and varying doses of alfentanil. Anesth Analg 1992; 75: 788–93.
Shiga T, Wajima Z, Inoue T, Sakamoto A. Predicting diffcult intubation in apparently normal patients: a meta-analysis of bedside screening test performance. Anesthesiology 2005; 103: 429–37.
Capron F, Alla F, Hottier C, Meistelman C, Fuchs-Buder T. Can acceleromyography detect low levels of residual paralysis? A probability approach to detect a mechanomyographic train-of-four ratio of 0.9. Anesthesiology 2004; 100: 1119–24.
Viby-Mogensen J, Engbaek J, Eriksson LI, et al. Good clinical research practice (GCrP) in pharmacodynamic studies of neuromuscular blocking agents. Acta Anaesthesiol Scand 1996; 40: 59–74.
Le Henanff A, Giraudeau B, Baron G, Ravaud P. Quality of reporting of noninferiority and equivalence randomized trials. JAMA 2006; 295: 1147–51.
Blackwelder WC “Proving the null hypothesis” in clinical trials. Control Clin Trials 1982; 3: 345–53.
Stout DM, Bishop MJ, Dwersteg JF, Cullen BF. Correlation of endotracheal tube size with sore throat and hoarseness following general anesthesia. Anesthesiology 1987; 67: 419–21.
Capan LM, Bruce DL, Patel KP, Turndorf H. Succinylcholine-induced postoperative sore throat. Anesthesiology 1983; 59: 202–6.
Doenicke A, Soukup J, Hoernecke R, Moss J. The lack of histamine release with cisatracurium: a double-blind comparison with vecuronium. Anesth Analg 1997; 84: 623–8.
Debaene B, Beaussier M, Meistelman C, Donati F, Lienhart A. Monitoring the onset of neuromuscular block at the orbicularis oculi can predict good intubating conditions during atracurium-induced neuromuscular block. Anesth Analg 1995; 80: 360–3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial support for this study was provided solely from institutional sources.
Conficts of interest: None declared.
Rights and permissions
About this article
Cite this article
Bouvet, L., Stoian, A., Jacquot-Laperrière, S. et al. Laryngeal injuries and intubating conditions with or without muscular relaxation: an equivalence study. Can J Anaesth 55, 674–684 (2008). https://doi.org/10.1007/BF03017743
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03017743