Abstract
Purpose
To examine the effects of temperature on auditory brainstem responses (ABRs) in infants during hypothermic cardiopulmonary bypass for total circulatory arrest (TCA). The relationship between ABRs (as a surrogate measure of corebrain temperature) and body temperature as measured at several temperature monitoring sites was determined.
Methods
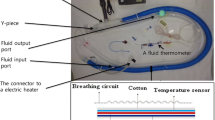
In a prospective, observational study, ABRs were recorded non-invasively at normothermia and at every 1 or 2°C change in ear-canal temperature during cooling and rewarming in 15 infants (ages: 2 days to 14 months) that required TCA. The ABR latencies and amplitudes and the lowest temperatures at which an ABR was identified (the threshold) were measured during both cooling and rewarming. Temperatures from four standard temperature monitoring sites were, simultaneously recorded.
Results
The latencies of ABRs increased and amplitudes decreased with cooling (P < 0.01), but rewarming reversed these effects. The ABR threshold temperature as related to each monitoring site (ear-canal, nasopharynx, esophagus and bladder) was respectively determined as 23 ± 2.2°C, 20.8 ± 1.7°C, 14.6 ± 3.4°C, and 21.5 ± 3.8°C during cooling and 21.8 ± 1.6°C, 22.4 ± 2.0°C, 27.6 ± 3.6°C, and 23.0 ± 2.4°C during rewarming. The rewarming latencies were shorter and Q10 latencies smaller than the corresponding cooling values (P < 0.01). Esophageal and bladder sites were more susceptible to temperature variations as compared with the ear-canal and nasopharynx.
Conclusion
No temperature site reliably predicted an electrophysiological threshold. A faster latency recovery during rewarming suggests that body temperature monitoring underestimates the effects of rewarming in the core-brain. ABRs may be helpful to monitor the effects of cooling and rewarming on the core-brain during pediatric cardiopulmonary bypass.
Résumé
Objectif
Examiner les effets de la température sur les réponses du tronc cérébral à des stimuli auditifs chez des enfants pendant la circulation extracorporelle hypothermique dans le cas d’un arrêt circulatoire total (ACT). La relation entre les réponses du tronc cérébral (en tant que mesure substitutive de la température cérébrale centrale) et la température du corps (mesurée à différents points d’enregistrement) a été déterminée.
Méthode
Lors d’une étude prospective d’observation, on a enregistré les réponses du tronc cérébral à des stimuli auditifs, de manière non effractive, à la température normale et à chaque changement de température de 1 ou 2 °C du canal auditif pendant le refroidissement et le réchauffement de 15 enfants (âgés de 2 jours à 14 mois) qui ont eu besoin d’ACT Les temps de latence et les amplitudes des réponses aux températures les plus basses auxquelles une réponse a été perçue (le seuil) ont été mesurés pendant le refroidissement et le réchauffement. Les températures de quatre points d’enregistrement standards ont été notées simultanément.
Résultats
Les temps de latence des réponses ont augmenté et les amplitudes ont diminué pendant le refroidissement (P < 0,01), mais le réchauffement a renversé ces effets. La température du seuil de réponse au canal auditif, au nasopharynx, à l’oesophage et à la vessie a été respectivement déterminée: 23 ± 2,2 °C, 20,8 ± 1,7 °C, 14,6 ± 3,4 °C, et 21,5 ± 3,8 °C pendant le refroidissement et 21,8 ± 1,6 °C, 22,4 ± 2,0 °C, 27,6 ± 3,6 °C, et 23,0 ± 2,4 °C pendant le réchauffement. Les temps de latence du réchauffement ont été plus courts et les temps de latence de Q10 plus faibles que les valeurs correspondantes du refroidissement (P < 0,01). L’oesophage et la vessie sont des points plus susceptibles aux changements de température en comparaison avec le canal auditif et la nasopharynx.
Conclusion
En aucun point d’enregistrement de la température on n’a pu prédire fidèlement un seuil électrophysiologique. Une récupération plus rapide du temps de latence pendant le réchauffement laisse croire que l’enregistrement de la température corporelle sous-évalue les effets du réchauffement central du cerveau. Les réponses du tronc cérébral aux stimuli auditifs peuvent servir à enregistrer les effets du refroidissement et du réchauffement central du cerveau pendant la circulation extracorporelle chez des enfants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Greeley WJ, Kern FH. Cerebral blood flow and metabolism during infant cardiac surgery. Paediatr Anaesth 1994; 4: 285–99.
Whitby JD, Dunkin LJ. Cerebral, oesophageal and nasopharyngeal temperatures. Br J Anaesth 1971; 43: 673–6.
Coselli JS, Crawford ES, Beall AC Jr, Mizrahi EM, Hess KR, Patel VM. Determination of brain temperatures for safe circulatory arrest during cardiovascular operation. Ann Thorac Surg 1988; 45: 638–42.
Stone JG, Young WL, Smith CR, et al. Do standard monitoring sites reflect true brain temperature when profound hypothermia is rapidly induced and reversed? Anesthesiology 1995; 82: 344–51.
Picton TW, Stapells DR, Campbell KB. Auditory evoked potentials from the human cochlea and brainstem. J Otolaryngol 1981; 10(Suppl9): 1–41.
Moller AR, Jannetta PJ. Neural generators of the auditory brainstem response.In: Jacobson JT (Ed.). The Auditory Brainstem Response. San Diego: College-Hill Press, 1985: 13–31.
Markand ON, Lee BI, Warren C, et al. Effects of hypothermia on brainstem auditory evoked potentials in humans. Ann Neurol 1987; 22: 507–13.
Kaga K, Takiguchi T, Myokai K, Shiode A. Effects of deep hypothermia and circulatory arrest on the auditory brain stem responses. Arch Otorhinolaryngol 1979; 225: 199–205.
Kusakari J, Inamura N, Sakurai T, Kawamoto K. Effect of hypothermia upon the electrocochleogram and auditory evoked brainstem response. Tohoku J Exp Med 1984; 143: 351–9.
Rodriguez RA, Audenaert SM, Austin EH III, Edmonds HL Jr. Auditory evoked responses in children during hypothermic cardiopulmonary bypass: report of cases. J Clin Neurophysiol 1995; 12: 168–76.
Chiappa KH. Brainstem auditory evoked potentials: methodology.In: Chiappa KH (Ed.). Evoked Potentials in Clinical Medicine. New York: Raven Press; 1983: 105–13.
Schwender D, Rimkus T, Haessler R, Klasing S, E. Pöppel, Peter K. Effects of increasing doses of alfentanil, fentanyl and morphine on mid-latency auditory evoked potentials. Br J Anaesth 1993; 71: 622–8.
Williston JS, Jewett DL. The Q10 of auditory brain stem responses in rats under hypothermia. Audiology 1982; 21: 457–65.
Baker MA, Stocking RA, Meehan JP. Thermal relationship between tympanic membrane and hypothalamus in conscious cat and monkey. J Appl Physiol 1972; 32: 739–12.
Stone JG, Schwartz AE, Finck AD, et al. Brain temperature monitoring during induced hypothermia. Anesthesiology 1995; 83: A173.
Nathan HJ, Lavallée G. The management of temperature during hypothermic cardiopulmonary bypass: I — Canadian survey. Can J Anaesth 1995; 42: 669–71.
Murkin JM. Hypothermic cardiopulmonary bypass — time for a more temperate approach? (Editorial) Can J Anaesth 1995; 42: 663–8.
Kern FH, Jonas RA, Mayer JE Jr, Hanley FL, Castaneda AR, Hickey PR. Temperature monitoring during CPB in infants: does it predict efficient brain cooling? Ann Thorac Surg 1992; 54: 749–54.
Bénita M, Condé H. Effects of local cooling upon conduction and synaptic transmission. Brain Res 1972; 36: 133–51.
Gold S, Cahani M, Sohmer H, Horowitz M, Shahar A. Effects of body temperature elevation on auditory nerve-brain-stem evoked responses and EEGs in rats. Electroenceph Clin Neurophysiol 1985; 60: 146–53.
Sohmer H, Gafni M, Chisin R. Auditory nerve-brain stem potentials in man and cat under hypoxic and hypercapnic conditions. Electroenceph Clin Neurophysiol 1982; 53: 506–12.
Guo J, Liao J-J, Preston JK, Batjer HH. A canine model of acute hidbrain ischemia and reperfusion. Neurosurgery 1995; 36: 986–93.
Sohmer H, Gafni M, Goitein K, Fainmesser P. Auditory nerve-brain stem evoked potentials in cats during manipulation of the cerebral perfusion pressure. Electroenceph Clin Neurophysiol 1983; 55: 198–202.
Thornton C, Catley DM, Jordan C, Royston D, Lehane JR, Jones JG. Enflurane increases the latency of early components of the auditory evoked response in man. Br J Anaesth 1981; 53: 1102–3.
Sokoloff L, Reivich M, Kennedy C, et al. The [14C] deoxyglucose method for the measurement of local cerebral glucose utilization: theory, procedure, and normal values in the conscious and anesthetized albino rat. J Neurochem 1977; 28: 897–916.
Author information
Authors and Affiliations
Corresponding author
Additional information
RAR was a Cadwell Laboratories Clinical Fellow and partially supported by the Alliant Community Trust. Monitors and accesories were provided by Cadwell Laboratories (USA)
Rights and permissions
About this article
Cite this article
Rodriguez, R.A., Edmonds, H.L., Auden, S.M. et al. Auditory brainstem evoked responses and temperature monitoring during pediatric cardiopulmonary bypass. Can J Anesth 46, 832–839 (1999). https://doi.org/10.1007/BF03012971
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03012971