Abstract
Purpose
The bradycardia produced by pyridostigmine and physostigmine in an animal model of acute cardiac denervation was examined according to its relation to cholinesterase inhibition and sensitivity to block by cholinergic receptor antagonists.
Methods
Cats were anaesthetised, vagotomised and propranolol-treated. Heart rate was continuously recorded. Erythrocyte cholinesterase activity of arterial blood was measured using a radiometric technique. Nicotinic and muscarinic M1 receptors were blocked with hexamethonium and pirenzepine, respectively. M2 receptors were blocked with gallamine, pancuronium and AFDX-116.
Results
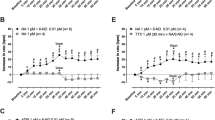
With pyridostigmine and physostigmine, the dose-response relationship for the decrease in heart rate (ED50 1.05 ± 0.25 and 0.198 ± 0.03 mg·kg−1, respectively) was shifted to the right of that for the inhibition of cholinesterase activity (ED50 0.094 ± 0.03 and 0.032 ± 0.01 mg·kg−1, respectively). The decrease in cholinesterase activity reached a plateau at a cumulative dose of 0.56 ± 0.08 and 0.32 ± 0.08 mg·kg−1, respectively. In contrast, there did not appear to be a plateau in the bradycardic effect. The bradycardia produced by pyndostigmine and physostigmine was blocked by hexamethonium (ED50 10 ± 1.3 and 15.3 ± 2.4 mg·kg−1, respectively), pirenzepine (ED50 68 ± 16 and 138 ± 32 μg·kg−1. respectively), gallamine (56 ± 11 and 67 ± 17 μg·kg−1, respectively ), pancuronium (32 ± 10 and 30 ± 4 μg·kg−1, respectively), and AFDX-116 (31 ± 4 and 28 ± 4 μg·kg−1, respectively).
Conclusion
The bradycardia produced by reversible anticholinesterase drugs containing a carbamyl group is not dearly related to the degree of cholinesterase activity, and has a low sensitivity to nicotinic and muscannic M1 and a high sensitivity to muscarinic M2 receptor antagonists.
Résumé
Objectif
Examiner si la bradycardie provoquée par la physostigmine et la pyndostigmine sur un modèle de dénervation cardiaque animal est en rapport avec l’inhibition de la cholinestérase et la sensibilité au bloc du récepteur cholinergique par ses antagonistes.
Méthodes
Des chats ont été anesthésiés, vagotomisés et traités au propanolol. Leur fréquence cardiaque a été enregistrée continuellement. Une technique radiométnque a permis de mesurer dans le sang artériel l’activité de la cholinestérase érythrocytaire. On a bloqué les récepteurs nicotiniques avec de l’hexaméthonium et les récepteurs muscariniques M1 avec de la pirenzépine. Les récepteurs M2 étaient bloqués avec de la gallamine, du pancuronium et de l’AFDX-116.
Résultats
Avec la pyridostigmine et la physostigmine, la relation dose-effet en rapport avec la baisse de la fréquence cardiaque (ED50 respective de 1,05 ± 0,25 et de 0,198 ± 0,03 mg·kg−1) a dévié vers la droite de celle de l’inhibition de l’activité cholinestérasique (ED50 respective de 0,094 ± 0,03 et 0,032 ± 0.01 mg·kg−1). La baisse de l’activité cholinestérasique a atteint un plateau à la dose cumulative respective de 0,56 ± 0,08 et 0,32 ± 0,08 mg·kg−1. Par contre, il ne semblait pas y avoir de plateau en ce qui concerne l’effet bradycardisant. La bradycardie provoquée par la pyridostigmine et la physostigmine était bloquée par l’héxaméthonium (ED50 respective de 10 ± 1,3 et de 15,3 ± 2,4 mg·kg−1), la pérenzépine (ED50 respective de 68 ± 16 et de 138 ± 32 μg·kg−1), la gallamine (respectivement 56 ± 11 et 67 ± 17 μg·kg−1), le pancuronium (respectivement 32 ± 10 et 30 ± 4 μg·kg−1) et l’AFDX-116 (respectivement 31 ± 4 et 28 ± 4 μg·kg−1).
Conclusion
La bradycardie provoquée par les anticholinestérases réversibles porteuses d’un groupe carbamyl n’a pas de relation certaine avec le degré d’activité cholinestérasique et est faiblement sensible aux antagonistes du récepteur nicotinique et muscarinique M1, et fortement sensible aux antagonistes du récepteur muscarinique M2.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hill M. Death after neostigmine injection. BMJ 1949; 2: 601.
MacIntosh RR. Death following injection of neostigmine. BMJ 1949; 1: 852.
Backman SB, Bachoo M, Polosa C. Mechanism of the bradycardia produced in the cat by the anticholinesterase neostigmine. J Pharm Exp Ther 1993; 265: 194–200.
Backman SB, Stein RD, Blank DW, Collier B, Polosa C. Different properties of the bradycardia produced by neostigmine and edrophonium in the cat. Can J Anaesth 1996; 43: 731–10.
Akaike A, Ikeda SR, Brookes N, Pascuzzo GJ, Rickett DL, Albuquerque EX. The nature of the interactions of pyridostigmine with the nicotinic acetylcholine receptorionic channel complex. Mol Pharmacol 1984; 25: 102–12.
Carpenter DO, Greene LA, Shain W, Vogel Z. Effects of eserine and neostigmine on the interaction of α-bungarotoxin withAplysia acetylcholine receptors. Mol Pharmacol 1976; 12: 999–1006.
Cooper JC, Gutbrod O, Witzemann V, Methfessel C. Pharmacology of the nicotinic acetylcholine receptor from fetal rat muscle expressed inXenopus oocytes. Eur J Pharmacol 1996; 309: 287–98.
Cox B, Lomas DM. The effects of eserine and neostigmine on the guinea-pig ileum and on ilial longitudinal muscle strips. J Pharm Pharmacol 1972; 24: 541–6.
Kuhlmann J, Okonjo KO, Maelicke A. Desensitization is a property of the cholinergic binding region of the nicotinic acetylcholine receptor, not of the receptorintegral ion channel. FEBS Lett 1991; 279: 216–8.
Okonjo KO, Kuhlmann J, Maelicke A. A second pathway of activation of theTorpedo acetylcholine receptor channel. Eur J Biochem 1991; 200: 671–7.
Pascuzzo GJ, Akaike A, Maleque MA, et al. The nature of the interactions of pyridostigmine with the nicotinic acetylcholine receptor-ionic channel complex. Mol Pharmacol 1984; 25: 92–101.
Sadoshima J-i, Tokutomi N, Akaike N. Effects of neostigmine and physostigmine on the acetylcholine receptor-ionophore complex in frog isolated sympathetic neurones. Br J Pharmacol 1988; 94: 620–4.
Schrattenholz A, Godavac-Zimmermann J, Schäfer H-J, Albuquerque EX, Maelicke A. Photoaffinity labeling ofTorpedo acetylcholine receptor by physostigmine. Eur J Biochem 1993; 216: 671–7.
Shaw K-P, Aracava T, Akaike A, Daly JW, Rickett DL, Albuquerque EX. The reversible cholinesterase inhibitor physostigmine has channel-blocking and agonist effects on the acetylcholine receptor-ion channel complex. Mol Pharmacol 1985; 28: 527–38.
Sherby SM, Eldefrawi AT, Albuquerque EX, Eldefrawi ME. Comparison of the actions of carbamate anticholinesterases on the nicotinic acetylcholine receptor. Mol Pharmacol 1985; 27: 343–8.
Slater NT, Filbert M, Carpenter DO. Multiple interactions of anticholinesterases withAplysia acetylcholine responses. Brain Res 1986; 375: 407–12.
Storch A, Schrattenholz A, Cooper JC, et al. Physostigmine, galanthamine and codeine act as ‘noncompetitive nicotinic receptor agonists’ on clonal rat pheochromocytoma cells. Eur J Pharmacol 1995; 290: 207–19.
Wachtel RE. Comparison of anticholinesterases and their effects on acetylcholine-activated ion channels. Anesthesiology 1990; 72: 496–503.
Flacke W, Gillis RA. Impulse transmissionvia nicotinic and muscarinic pathways in the stellate ganglion of the dog. J Pharmacol Exp Ther 1968; 163: 266–76.
Hammer R, Giachetti A. Muscarinic receptor subtypes: M1 and M2 biochemical and functional characterization. Life Sci 1982; 31: 2991–8.
Shibata O, Kanairo M, Zhang S, et al. Anticholinesterase drugs stimulate phosphatidylinositol response in rat tracheal slices. Anesth Anaig 1996; 82: 1211–1.
Stainback R. Orthotopic cardiac transplantation: a model of the denervated heart.In: Robertson D, Biaggioni I (Eds.). Disorders Of The Autonomic Nervous System. Luxembourg: Harwood Academic Publishers, 1995: 335–74.
Backman SB, Ralley FE, Fox GS. Neostigmine produces bradycardia in a heart transplant patient. Anesthesiology 1993; 78: 777–9.
Backman SB, Fox GS, Stein RD, Ralley FE. Neostigmine decreases heart rate in heart transplant patients. Can J Anaesth 1996; 43: 373–8.
Backman SB, Stein RD, Ralley FE, Fox GS. Neostigmine-induced bradycardia following recent vs remote cardiac transplantation in the same patient. Can J Anaesth 1996; 43: 394–8.
Backman SB, Stein RD, Fox GS, Polosa C. Heart rate changes in cardiac transplant patients and in the denervated cat heart after edrophonium. Can J Anaesth 1997; 44: 247–54.
Allen TGJ, Burnstock G. M1 and M2 muscarinic receptors mediate excitation and inhibition of guinea-pig intracardiac neurones in culture. J Physiol 1990; 422: 463–80.
Author information
Authors and Affiliations
Additional information
This work was supported by grants from the Canadian Anaesthetists’ Society and the Royal Victoria Hospital Research Institute to SBB and from the Medical Research Council to BC. Preliminary results presented in part at the CAS 1994 Annual Meeting in Edmonton, Alberta).
An erratum to this article is available at http://dx.doi.org/10.1007/BF03012039.
Rights and permissions
About this article
Cite this article
Stein, R.D., Backman, S.B., Collier, B. et al. Bradycardia produced by pyridostigmine and physostigmine. Can J Anaesth 44, 1286–1292 (1997). https://doi.org/10.1007/BF03012778
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03012778