Abstract
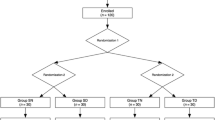
Alfentanil, a congener of the opioid fentanyl, possesses properties that make it an attractive choice for use during short operative procedures. Since the phannacodynamic aspects of alfentanil have not been well documented in children, this study was undertaken to evaluate the safety, efficacy, and dose requirements of alfentanil when used with nitrous oxide or halothane in paediatric patients. Eighty unpremedicated patients, ASA physical status I or II and aged 2–12 yr were studied. Patients were randomly assigned to one of four groups. After induction of anaesthesia with nitrous oxide, oxygen, and halothane, the groups were treated as follows. In Group I (n = 19), after halothane was discontinued, alfentanil 50 μg · kg−1 was infused over 30 sec. In Group 2 (n = 20), the end-tidal halothane was maintained at 0.5% and alfentanil 25 μg · kg−1 was infused. In Group 3 (n = 20), the end-tidal halothane concentration was maintained at 1% and alfentanil 12.5 μg · kg−1 was infused. In Group4(n = 21), the end-tidal halothane concentration was maintained at 1.5% and no alfentanil was administered. Patients in Groups 1, 2, and 3 received bolus doses of alfentanil 12.5 μg · kg−1 as needed to maintain haemodynamic stability. After alfentanil administration, there were transient decreases in systolic blood pressure in Groups 1 and 2, and in heart rate in Group 2. With surgical stimulation, haemodynamic stability was well maintained except in patients in Group 1, who had an increase in systolic blood pressure. Children Group 1 were alert sooner and their tracheas were extubated earlier than those in Groups 2, 3, and 4. The four groups were similar in postoperative narcotic analgesic administration and incidence of vomiting. In summary, alfentanil (12.5–50.0 μg · kg−1) was a safe anaesthetic, whether combined with nitrous oxide alone or with nitrous oxide and halothane.
Résumé
On connait mal la pharmacocinétique de l’alfentanil chcz les enfants même si certaines des propriétés de ce parent dufentanyl devraient en favoriser l’usage tors de courtes interventions. Nous avons done évalué chez 80 enfants la sûreté et iefficacité de diverses combinaisons d’alfentanil et d’halothane en presence de protoxyde d’azote. On randomisait d’abord les enfants de 2 à 12 ans et de classe ASA 1 ou 2 en quatre groupes. Sans prémédication, on induisait l’anesthésie de façon habituelle avec de l’oxygène, du protoxyde d’azote et de l’halothane. Puis, on cessait l’halothane chez les enfants du groupe 1 (n = 19) pour leur injecter 50 μg · kg−1 d’alfentanil en 30 secondes. Pour le groupe 2 (n = 20), on ajustait l’halothane enfin d’expiration à 0,5% et on y ajoulait 25 μg · kg−1 d’alfentanil. Le groupe 3 (n = 20) avail une combinaison d’halothane 1,0% et 12,5 μg · kg−1 d’alfentanil et enfin, le groupe 4 (n = 21) n’avail que 1,5% d’halothane sans alfentanil. On injectait subséquemment des dosesde 12,5 μg · kg−1 d’alfentanil aicx enfants des groupes 1, 2 el 3 afin dassurer la stabilité hémodynamique. Transitoirement, avec l’injection d’alfentanil, la tension artérielle systolique diminua dans les groupes 1 et 2 et le pouls ralentit dans le groupe 2. Il n’y eut pas de changement hémodynamique associée à la stimulation chirurgicale sauf dans le groupe 1 ou la tension artérielle systolique s’éleva. Les enfants du groupe 1 s’éveillèrent plus rapidement et purenl être extubes plus tôt que ceux des autres groupes. En postopératoire, les besoins en analgésiques el l’incidence de vomissemenl furent semblables chez les quatre groupes. Bref, en doses de 12,5 à 50 μg · kg−1, l’alfentanil associé an protoxyde d’azote constitue tin anesthésique sûr, qu’il soit ou non combine avec de l’halothane.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bovill JG, Sebel PS, Blackburn CL, Heykants J. The pharmacokinetics of alfentanil (R39209): a new opioid analgesic. Anesthesiology 1982; 57: 439–42.
Nauta J, de Lange S, Koopman D, Spierdijk J, VanKleff J, Stanley TH. Anesthetic induction with alfentanil: a new short-acting narcotic analgesic. Anesth Analg 1982; 61: 267–72.
Ausems ME, Hug CC, de Lange S. Variable rate infusion of alfentanil as a supplement to nitrous oxide anesthesia for general surgery. Anesth Analg 1983; 62: 982–6.
Meistelman C, Saint-Maurice C, Lepaul M, Levron JC, Loose JP, MacGee K. A comparison of alfentanil pharmacokinetics in children and adults. Anesthesiology 1987; 66: 13–6.
Kay B, Pleuvey B. Human volunteer studies of alfentanil (R 39209), a new short acting narcotic analgesic. Anaesthesia 1980; 35: 952–6.
Kramer M, Kling D, Waiter P, Borman B, Heneplema G. Alfentanil, a new short acting opioid: hemodynamic and respiratory aspects. Anaesthetist 1983; 32: 265–71.
Marlow N, Weindling AM, Cooke RW. Hazards of analgesia for newborn infants. Arch Dis Child 1988; 63: 1293.
Davis PJ, Killian A, Stiller RL, Cook DR, Guthrie RD, Scierka AM. Pharmacokinetics of alfentanil in newborn premature infants and older children. Dev Pharmacol Ther 1989; 13: 21–7.
Ausems ME, Hug CC Jr, Stanski DR, Burm AG. Plasma concentrations of alfentanil required to supplement nitrous oxide anesthesia for general surgery. Anesthesiology 1986; 65: 362–73.
White PF, Coe V, Shafer A, Sung ML. Comparison of alfentanil with fentanyl for outpatient anesthesia. Anesthesiology 1986; 64: 99–106.
Cartwhght DP. Recovery after anaesthesia with alfentanil or halothane. Can Anaesth Soc J 1985; 32: 479–83.
Short SM, Rutherfoord CF, Sebel PS. A comparison between isoflurane and alfentanil supplemented anaesthesia for short procedures. Anaesthesia 1985; 40: 1160–4.
Borland LM, Saitz EW, Woelfel SK. Evaluation of pcdiatric anesthesia care. Anesthesiology 1989; 71: A920.
Steward DJ. Experiences with an outpatient anesthesia service for children. Anesth Analg 1973; 52: 877–80.
Patel RI, Hannallah RS. Anesthetic complications following pediatric ambulatory surgery: a 3-yr study. Anesthesiology 1988; 69: 1009–12.
Christensen S, Farrow-Gillespie A, Lennan J. Incidence of emesis and postanesthetic recovery after strabismus surgery in children: a comparison of droperidol and lidocaine. Anesthesiology 1989: 70: 251–4.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mulroy, J.J., Davis, P.J., Rymer, D.B. et al. Safety and efficacy of alfentanil and halothane in paediatric surgical patients. Can J Anaesth 38, 445–449 (1991). https://doi.org/10.1007/BF03007581
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03007581