Abstract
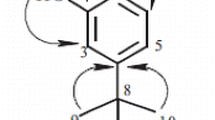
The fruits ofMorinda citrifolia, Linn. afforded a new constituent, morinaphthalenone (1), and three known constituents, scopoletin (2), 1, 3-dimethoxy-anthraquinone (3) and 1, 2-dihydroxy-anthraquinone (4). The structures of these isolated compounds were determined by spectroscopic methods, including 1D- and 2D-NMR (COSY-45, HMQC, HMBC) techniques, as well as by comparison with published values.
Similar content being viewed by others
References
Bargues, V., Blay, G., Garcia, B., Garcia, C. L., and Pedro, J. R., Synthesis of 9-Oxyfunctionalized Eudesmanes from artemisin.Tetrahedron, 51, 5609–5616 (1995).
Bohlmann, F. and Jakupovic, J., 8-Oxo-a-Selinen und neue scopoletin-derivate ausConyza-arten.Phytochemistry, 18, 1367–1370 (1979).
Cardona, L., Garcia, B., Pedro, J. R., and Perez, J., 6-Prenyloxy- 7-Methoxy-coumarin, A coumarin-hemiterpene ether fromCarduus tenuiflorus.Phytochemistry, 31, 3989–3991 (1992).
Fraga, B. M., Hernandez, M. G., Mestres, T., Arteaga, J. M., and Perales, A., Eudesmane Sesquiterpenes fromTeucrium heterophyllum. The X-ray structure of Teucdiol A.Phytochemistry, 34, 1083–1086 (1993).
Hill, R. A., Krebs, H. C., Verpoorte, R., and Wijnsma, R., Progress in the Chemistry of Organic Natural Products, Springer-Verlag Wien New York, vol. 49, pp. 79–149 (1986).
Inoue, K., Shiobara, Y., Nayeshiro, H., Inouye, H., Wilson, G., and Zenk, M. H., Biosynthesis of anthraquinones and related compounds inGalium mollugo cell suspension cultures.Phytochemistry, 23, 307–311 (1984).
Iwagawa, T. and Hase, T., A coumarin acetylglucoside fromViburnum suspen-sum.Phytochemistry, 23, 467–468 (1984).
Luc, L., (S.A. Nigiko), Insecticidal fatty acids fromMorinda fruits. Fr. Demande FR 2,673,639 (Cl. CIIB1/10), 11 sep 1992, Appl. 91/2,859, 08 Mar 1991; 18 pp.
Mulder-Krieger, T., Verpoorte, R., Water, A., Gessel, M. V., Oeveren, B. C. J. A. V., and Svendsen, A. B., Identification of the alkaloids and anthraquinones inCinchona ledgeriana callus cultures.Planta Med., 46, 19–24 (1982).
Muzychkina, R. A.,Natural Anthraquinones, Phasis Research and Publishing Corporation 42-44. presnenski val 123557 Moscow, Russia (1998).
Nakanishi, K. and Solomon, P. H., Infrared Absorption Spectroscopy, Holden-day, Inc., 500 Sansome Street, San Francisco, Ca, Second edition, p. 14 (1977).
Rusia, K. and Srivastava, S. K., A new anthraquinone from the roots ofMorinda citrifolia Linn.Curr. Sci., 58, 249 (1989).
Saludes, J. P., Garson, M. J., Franzblau, S. G., and Aguinaldo, A. M., Antitubercular constituents from the hexane fraction ofMorinda citrifolia linn.Phytother. Res., 16, 683–685 (2002).
Scott, A. I., Interpretation of the Ultraviolet Spectra of Natural Products, Pergamon Press, Oxford, London, Edinburgh, New York, Paris, Frankfurt, pp. 286–288 (1964).
Sibanda, S., Ndengu, B., Multari, G., Pompi, V., and Galeffi, C., A courmarin glucoside fromXeromphis obovata.Phytochemistry, 28, 1550–1552 (1989).
Sugama, K. O., Hayashi, K., and Mitsuhashi, H. O., Eremophi- lenolides fromPetasites japonicus.Phytochemistry, 24, 1531–1535 (1985).
Wang, M.-Y, West, B. J., Jensen C. J., Nowicki, D., Su, C., Palu, A. K., and Anderson, G.,Morinda citrifolia (Noni): a literature review and recent advances in Noni research.Acta Pharmacol. Sin., 23, 1127–1141 (2002).
Williams, D. H. and Fleming, I., Spectroscopic Methods in Organic Chemistry, Tata McGraw- Hill Publishing Company Limited New Delhi, pp. 97–98, (1988).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Siddiqui, B.S., Sattar, F.A., Ahmad, F. et al. Isolation and structural elucidation of chemical constituents from the fruits ofMorinda citrifolia Linn. Arch Pharm Res 30, 919–923 (2007). https://doi.org/10.1007/BF02993956
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02993956