Abstract
Background
Organic pollutants formed during thermal treatment of oil shale and then released from the solid waste (semicoke) to aquatic life are a major concern in Estonia. Efficient environmentally friendly techniques are being sought for the analysis of soil and sediments for pollutants and for the clean up of contaminated areas. The altered physico-chemical properties of pressurised hot water can be exploited in the extraction of organics from solid samples. For example, the relative permittivity, hydrogen bonding ability and viscosity of water are decreased and diffusivity is increased with temperature. In addition, water is environmentally friendly, cheap, non-flammable and readily available. In small-scale or pilot-scale operations, pressurised hot water extraction (PHWE) can also be used in the purification of contaminated soil and sediments.
Objective
PHWE and conventional Soxhlet extraction were applied to extract organic compounds from soil and sediment samples collected from various locations around a semi-coke mound in a mining district in northeastern Estonia. One important aim was to compare the extraction efficiencies of the two techniques. Another aim was to determine the pollutants in soil in the vicinity of the semi-coke mound and the sediments of two rivers (the Purtse River flowing to the Gulf of Finland and the Kohtla River feeding into the Purtse) and in canals between the Kohtla River and the semi-coke mound.
Methods
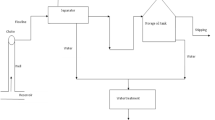
The PHWE equipment was self-constructed and applied with temperatures of 300 and 350°C (P = ca. 200 bar). Soxhlet extraction was carried out for 20 h with dichloromethane as a solvent. All extracts were cleaned up with a silica gel column, concentrated and analysed by gas chromatography-mass spectrometry (GC-MS). Organic matter contents of the samples were determined.
Results and Discussion
Alkanes and polyaromatic hydrocarbons (PAHs) were the main compounds found in the GC-MS analysis after.PHWE and Soxhlet extractions. In general, recoveries for short-chainn-alkanes (C10-C16) and PAHs with two benzene rings (i.e. naphthalene, acenaphthylene, acenaphthene and fluorene) were better with PHWE at 350°C than with Soxhlet extraction. These compounds are relatively volatile and may be lost during Soxhlet extraction. For longer chainn-alkanes, recoveries were better with Soxhlet extraction than with PHWE. The longer chain compounds are less polar than the shorter chain compounds and not so easily recovered by PHWE. For PAHs with more than two benzene rings, PHWE at 350°C produced roughly similar recoveries as Soxhlet extraction. PHWE extraction efficiency forn-alkanes was better at 350°C than at 300°C due to the lower polarity and increased diffusivity of water and better thermal desorption of the compounds at higher temperatures. For the same reasons, PHWE was generally more efficient at 350°C than at 300°C for PAHs, especially for the compounds with highest molar masses. Organic matter content was the greatest in the samples with high PAH and alkane concentrations, showing that organic matter is a good sink for hydrophobic micropollutants. The highest concentrations ofn-alkanes (maximum individual alkane concentrations ca. 8 µg/g) and PAHs (maximum individual PAH concentrations ca. 20 µg/g) were found in the soil under the semi-coke mound, showing that the compounds were flushed down with water. Almost concentrations ofn-alkanes and PAHs as high as under the mound were found within the mound and in sediment from the canal ca. 10 m from the mound. Sediment from the Kohtla River where it meets the Purtse River, ca. 14 km from the semi-coke mound, was also contaminated with the compounds. Only somen-alkanes were found at the mouth of the Purtse River indicating that the major part of the pollutants were accumulated in the sediments of the Kohtla River.
Conclusions
The PHWE equipment was reliable and safe to use. PHWE produced better recoveries for short-chain alkanes (C10-C16) and PAHs with two benzene rings than did Soxhlet extraction, but it was less efficient than Soxhlet for larger compounds. The two extraction methods together thus provide a good tool for extractingn-alkanes and PAHs. PAH and alkane concentrations were the most abundant in the samples with high organic matter content. The samples collected near the semi-coke mound and in the Kohtla River were contaminated withn-alkanes and PAHs, but only somen-alkanes were found in the Purtse River, showing that the heaviest contamination of soil and sediments was localised relatively close to the semi-coke mound.
Recommendations and Outlook
With PHWE, the hazardous organic solvents used in extraction processes can be replaced with water. Increasing the volume of the extraction vessel and applying a more efficient heating system and higher flow rate would enable the use of PHWE on a pilot-scale in the remediation of contaminated soil and sediments. With the present equipment, the PHWE-treated samples can also be oxidised directly on-line under supercritical conditions by feeding oxidant to the reactor situated after the extraction vessel. Organics can be completely destroyed with water and carbon monoxide, the main reaction products.
Similar content being viewed by others
References
Estonian Environment Information Centre (2001): State of environment in Estonia — on the threshold of XXI century, Tallinn, 96 pp
Kundel H, Liblik V (2000): Emission of volatile phenols from stabilization ponds of oil shale ash dump leachate. Oil Shale 17, 81–94
Huuskonen SE, Tuvikene A, Trapido M, Fent K, Hahn M (2000): Cytochrome P4501A induction and porphyrin accumulation in PLHC-1 cells exposed to sediment and oil shale extracts. Arch Environ Contam Toxicol 38, 59–69
US-EPA, Code of Federal Regulation, Title 40, Part 60, subparts D, Da, Db, Dc; Environmental Protection Agency: Washington DC, 1997, p 44
Jacob J (1996): The significance of polycyclic aromatic hydrocarbons as environmental carcinogens. Pure Appl Chem 68, 301–308
Hyötyläinen T, Oikari A (1999): The toxicity and concentrations of PAHs in creosote-contaminated lake sediment. Chemosphere 38, 1135–1144
McGowan TF, Greer BA, Lawless M (1996): Thermal treatment and non-thermal technologies for remediation of manufactured gas plant sites. Waste Manage. 16, 691–698
Peterson EJ, Spall WD (1983): Physical and chemical characterization and comparison of solids, liquids, and oil from Estonian and Green River formation shales. Los Alamos National Laboratory report, LA-9722-MS
Hawthorne SB, Grabanski CB, Martin E, Miller DJ (2000): Comparisons of Soxhlet extraction, pressurized liquid extraction, supercritical fluid extraction and supercritical water extraction for environmental solids: recovery, selectivity and effects on sample matrix. J Chromatogr A 892, 421–433
Hartonen K, Meissner G, Kesälä T, Riekkola M-L (2000): Pressurized hot water extraction (PHWE) ofn-alkanes and polyaromatic hydrocarbons (PAHs): Comparison for PAHs with supercritical fluid extraction. J Microcolumn Sep 12, 412–418
Li W, Lazar IM, Wan YJ, Butala SJ, Shen Y, Malik A, Lee ML (2000): Determination of volatile hydrocarbons in coals and shales using supercritical fluid extraction and chromatography. Energy Fuels 11, 945–950
Koel M, Ljovin S, Bondar E (2000): Supercritical carbon dioxide extraction of Estonian oil shale. Oil Shale 17, 225–232
Hartonen K, Bøwadt S, Hawthorne S B, Riekkola M-L (1997): Supercritical fluid extraction with solid-phase trapping of chlorinated and brominated pollutants from sediment samples. J Chromatogr A 774, 229–242
Hawthorne SB, Yang Y, Miller DJ (1994): Extraction of organic pollutants from environmental solids with sub- and supercritical water. Anal Chem 66, 2912–2920
Yang Y, Hawthorne SB, Miller DJ (1997): Class-selective extraction of polar, moderately polar, and nonpolar organics from hvdrocarbon wastes using subcritical water. Environ Sci Technol 31, 430–437
Lagadec AJM, Miller DJ, Lilke AV, Hawthorne SB (2000): Pilot-scale subcritical water remediation of polycyclic aromatic hydrocarbon- and pesticide-contaminated soil. Environ Sci Technol 34, 1542–1548
Andersson T, Hartonen K, Hyötyläinen T, Riekkola M-L (2002): Pressurised hot water extraction and thermal desorption of polycyclic aromatic hydrocarbons from sediment with use of a novel extraction vessel. Anal Chim Acta 446, 93–100
Yang Y, Bøwadt S, Hawthorne SB, Miller DJ (1995): Subcritical water extraction of polychlorinated biphenyls from soil and sediment. Anal Chem 67, 4571–4576
Hawthorne SB, Grabanski CB, Hageman KJ, Miller DJ (1998): Simple method for estimating polychlorinated biphenyl concentrations on soils and sediments using subcritical water extraction coupled with solid-phase microextraction
van Bavel B, Hartonen K, Rappe C, Oberg L, Riekkola M-L (1999): Pressurised hot water extraction (PHWE) of PCDFs from industrial soil. Organohalogen Compd 40, 577–580
van Bavel B, Hartonen K, Rappe C, Riekkola M-L (1999): Pressurised hot water/steam extraction of polychlorinated dibenzofurans and naphthalenes from industrial soil. Analyst 124, 1351–1354
Ogunsola O, Berkowitz N (1995): Extraction of oil shales with sub- and near-critical water. Fuel Process. Technol. 45, 95–107
Franck EU (1970): Water and aqueous solutions at high pressures and temperatures. Pure Appl Chem 24, 13–30
Dudziak KH (1966): Messungen der Viskosität des Wassers bis 560°C und 3500 bar. Ber Bunsenges Phys Chem 70, 1120–1128
Khan A (2000): A liquid water model: density variation from supercooled to superheated states, prediction of H-bonds, and temperature limits. J Phys Chem B 104, 11268–11274
Hoffmann MM, Conradi MS (1997): Are there hydrogen bonds in supercritical water? J Am Chem Soc 119, 3811–3817
Yoshii N, Miura S, Okazaki S (2001): A molecular study of dielectric constant of water from ambient to sub- and supercritical conditions using a fluctuating-charge potential model. Chem Phys Lett 345, 195–200
Uematsu M, Franck EU (1980): Static dielectric constant of water and steam. J Phys Chem Ref Data 9, 1291–1305
Weast RC, Astle MJ, Beyer WH (1987) (eds): CRC Handbook of Chemistry and Physics, 67th ed., CRC Press Inc, Florida USA
Miller DJ, Hawthorne SB, Gizir AM, Clifford AA (1998): Solubility of polycyclic aromatic hydrocarbons in subcritical water from 298 K to 498 K. J Chem Eng Data 43, 1043–1047
Andersson T, Hartonen K, Hyotylainen T, Riekkola M-L (2003): Stability of polycyclic aromatic hydrocarbons in pressurised hot water. Analyst 128, 150–155
Kronholm J, Kalpala J, Hartonen K, Riekkola M-L (2002): Pressurized hot water extraction coupled with supercritical water oxidation in remediation of sand and soil containing PAHs. J Supercrit Fluids 23, 123–134
Kronholm J, Desbands B, Hartonen K, Riekkola M-L (2002): Environmentally friendly laboratory-scale remediation of PAH-contaminated soil by using pressurized hot water extraction coupled with pressurized hot water oxidation. Green Chem 4, 213–219
Truu, J, Heinaru E, Talpsep E, Heinaru A (2002): Analysis of river pollution data from low-flow period by means of multivariate techniques, a case study from the oil-shale industry region, northeastern Estonia. ESPR — Environ Sci & Pollut Res, Special Issue 1, 8–14
Alonso MC, Puig D, Silgoner I, Grasserbauer M, Barceló D (1998): Determination of priority phenolic compounds in soil samples by various extraction methods followed by liquid chromatography-atmospheric pressure chemical ionisation mass spectrometry. J Chromatogr A 823, 231–239
Hawthorne SB, Grabanski CB (2000): Vaporization of polyaromatic hydrocarbons (PAHs) from sediments at ambient conditions. Environ Sci Technol 34, 4348–4353
Hawthorne SB, Poppendieck DG, Grabanski CB, Loehr RC (2001): PAH release during water desorption, supercritical carbon dioxide extraction, and field bioremediation. Environ Sci Technol 35, 4577–4583
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kronholm, J., Kettunen, J., Hartonen, K. et al. Pressurised hot water extraction ofn-alkanes and polyaromatic hydrocarbons in soil and sediment from the oil shale industry district in estonia. J Soils & Sediments 4, 107–114 (2004). https://doi.org/10.1007/BF02991054
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02991054