Abstract
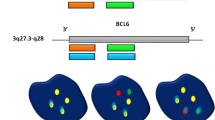
Follicular lymphoma is categorized as low-grade lymphoma because of the long median survival, but the disease is difficult to cure because of frequent recurrence. The t(14;18)(q32;q21) associated with follicular lymphoma juxtaposes a portion of BCL-2 (18q21) and IGH(14q32); the result is bcl-2 overexpression. In this study, a highly sensitive 2-color fluorescence in situ hybridization (FISH) method was used to detect t(14;18)(q32;q21) in the nuclei of paraffin-embedded tissue sections. Fourteen specimens, including 11 samples from follicular lymphoma and 3 samples from diffuse large cell lymphoma, for which results of karyotype study and paraffin-embedded tissues were available were selected for FISH study. The FISH results were compared with results of karyotype study of the lymph nodes involved in lymphoma. Among our 11 patients with the diagnosis of follicular lymphoma in whom karyotype study was performed, 8 had t(14;18)(q32;q21) in karyotype analysis, and 7 of these patients had a positive pattern in FISH analysis. In 1 case, FISH analysis was difficult because of weak signals. All 3 patients with diffuse large cell lymphoma and t(14;18)(q32;q21) in karyotype analysis had a positive pattern in FISH analysis. In 3 cases of follicular lymphoma without t(14;18)(q32;q21) in karyotype analysis, FISH did not show a positive pattern. Therefore the FISH assay in tissue was found to be very sensitive in detection of IGH/BCL2 translocation and was helpful in diagnosis of follicular lymphoma or in clarification of the cell origin of lymphoma when karyotype analysis was not available. Performing FISH on paraffin sections also is useful because we can identify cells with genetic abnormalities in the tumor and make a retrospective cytogenetic diagnosis even with old paraffin-embedded specimens. Int J Hematol. 2003;78:154-159.
Similar content being viewed by others
References
Greer JP, Macon WR, McCurley TL. Follicular center cell lymphomas. In: Lee GR, Foerster J, Lukens J, et al, eds.Wintrobe’s Clinical Hematology. 10th ed. Baltimore:Williams & Wilkins; 1999:2472–2474.
Tsujimoto Y, Jaffe E, Cossman J, Gorham J, Nowell PC, Croce CM. Clustering of breakpoints on chromosome 11 in human B-cell neoplasms with the t(11;14) chromosome translocation.Nature. 1985;315:340–343.
Tsujimoto Y, Croce CM. Analysis of the structure, transcripts, and protein products of bcl-2, the gene involved in human follicular lymphoma.Proc Natl Acad Sci U S A. 1986;83:5214–5218.
Cleary ML, Smith SD, Sklar J. Cloning and structural analysis of cDNAs for bcl-2 and a hybrid bcl-2/immunoglobulin transcript resulting from the t(14;18) translocation.Cell. 1986;47:19–28.
McKeithan TW. Molecular biology of non-Hodgkin’s lymphomas.Semin Oncol. 1990;17:30–42.
Erikson J, Finan J, Tsujimoto Y, Nowell PC, Croce CM. The chromosome 14 breakpoint in neoplastic B cells with the t(11;14) translocation involves the immunoglobulin heavy chain locus.Proc Natl Acad Sci U S A. 1984;81:4144–4148.
Macintyre EA, Delabesse E. Molecular approaches to the diagnosis and evaluation of lymphoid malignancies.Semin Hematol. 1999; 36:373–389.
Rack KA, Salomon-Nguyen F, Radford-Weiss I, et al. FISH detec tion of chromosome 14q32/IgH translocations: evaluation in follicular lymphoma.Br J Haematol. 1998;103:495–504.
Nathwani BN, Piris MA, Harris NL, et al. Follicular lymphoma. In: Jaffe ES, Harris NL, Stein H, Vardiman JW, eds.World Health Organization Classification of Tumours: Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. Lyon, France: IARC Press; 2001:162–167.
Vaandrager JW, Schuuring E, Kluin-Nelemans HC, Dyer MJS, Raap AK, Kluin PM. DNA fiber fluorescence in situ hybridization analysis of immunoglobulin class switching in B-cell neoplasia: aberrant CH gene rearrangements in follicle center-cell lymphoma.Blood. 1998;92:2871–2878.
Remstein ED, Kurtin PJ, Buno I, et al. Diagnostic utility of fluorescence in situ hybridization in mantle-cell lymphoma.Br J Haematol. 2000;110:856–862.
Huang JZ, Sanger WG, Greiner TG, et al. The t(14;18) defines a unique subset of diffuse large B-cell lymphoma with a germinal center B-cell gene expression profile.Blood. 2002;99:2285–2290.
Zhang Y, Cheung ANY, Chan ACL, et al. Detection of trisomy 3 in primary gastric B-cell lymphoma by using chromosome in situ hybridization on paraffin sections.Am J Clin Pathol. 1996;9:995–1000.
Blanco R, Lyda M, Dabis B, Kraus M, Fenoglio-Preiser C. Trisomy 3 in gastric lymphomas of extranodal marginal zone B-cell (mucosa-associated lymphoid tissue) origin demonstrated by FISH in intact paraffin tissue sections.Hum Pathol. 1999;30:706–711.
Nomura K, Sekoguchi S, Ueda K, et al. Differentiation of follicular from mucosa-associated lymphoid tissue lymphoma by detection of t(14;18) on single-cell preparations and paraffin-embedded sections.Gene Chromosome Cancer. 2002;33:213–216.
Nomura K, Yoshino T, Nakamura S, et al. Detection of t(11;18) (q21;q21) in marginal zone lymphoma of mucosa-associated lymphatic tissue type on paraffin-embedded tissue sections by using fluorescence in situ hybridization.Cancer Genet Cytogenet. 2003; 140:49–54.
Horsman DE, Gascoyne RD, Coupland RW, et al. Comparison of cytogenetic analysis, Southern analysis, and polymerase chain reaction for the detection of t(14;18) in follicular lymphoma.Am J Clin Pathol. 1998;103:472–478.
Weiss LM, Warnke RA, Sklar J, et al. Molecular analysis of the t(14;18) chromosomal translocation in malignant lymphoma.N Engl J Med. 1987;317:1185–1189.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hirose, Y., Masaki, Y. & Ozaki, M. Fluorescence In Situ Hybridization Detection of Chromosome IGH/BCL2 Translocations from Paraffin-Embedded Tissue: Evaluation in Follicular Lymphoma. Int J Hematol 78, 154–159 (2003). https://doi.org/10.1007/BF02983385
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02983385