Abstract
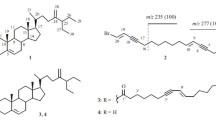
Two new sulfated saponins designated as certonardosides P2 and l3 (1 and2) were isolated from the brine shrimp active fraction of the MeOH extract of the starfishCertonardoa semiregularis. The structures were determined on the basis of spectral analysis. Compounds1 and2 were tested for cytotoxicity against five human tumor cell lines (A549, SK-OV-3, SK-MEL-2, XF498, and HCT15), and compound 1 displayed significant cytotoxicity against the SK-MEL-2 skin cancer cell.
Similar content being viewed by others
References
Anastasia, M., Allevi, P., Ciuffreda, P., and Riccio R., Configuration assignment of 24R-and 24S-isomers of 29-oxygenated steroids by1H- and13C-NMR spectroscopy.Tetrahedron, 42, 4843–4847(1986).
Chludil, H. D., Seldes, A. M., and Maier, M. S., Antifungal steroidal glycosides from the patagonian starfishAnasterias minuta: Structure-activity correlations.J. Nat. Prod., 65, 153–157(2002).
D' Auria, M. V., Minale L., and Riccio, R., Polyoxygenated steroids of marine origin.Chem. Rev., 93,1839–1898 (1993).
De Marino, S., lorizzi, M., Palagiano, E., Zollo, F., and Roussakis, C., Isolation, structure elucidation, and biological activity of the steroid oligoglycosides from an antarctic starfish of the familyAsteriidae.J. Nat. Prod., 61, 1319–1327 (1998).
Eggert, H., Van Antwerp, C. L., Bhacca, N. B., and Djerassi, C., Carbon-13 nuclear magnetic resonance spectra of hydroxyl steroids.J. Org. Chem., 41,71–78 (1976).
lorizzi, M., Minale, L., Riccio, R., Debray, M., and Menou, J. L., Starfish saponins, part 23. Steroidal glycosides from the starfishHalityle regularis.J. Nat. Prod., 49,67–78 (1986).
lorizzi, M., De Marino, S., and Zollo, F., Steroidal oligoglycosides from the Asteroidea.Curr. Org. Chem., 5, 951–973 (2001).
Ivanchina, N. V., Kicha, A. A., Kalinovsky. A. I., Dmitrenok, P. S., Stonik, V. A., Riguera, R., and Jimenez, C., Hemolytic polar steroidal constituents of the starfishAphelasterias japonica.J. Nat. Prod., 63,1178–1181 (2000).
Minale, L., Pizza, C., Plomitallo, A., Riccio, R., Zollo, R., and Mellon, R. A., Starfish saponins. XII. Sulfated steroid glycosides from the starfishHacelia attenuata.Gazz. Chim. Ital., 114,151–158(1984).
Minale L., Riccio, R., and Zollo, R., Structural studies on chemical constituents of echinoderms.Stud. Nat. Prod. Chem., 15,43–110(1995).
Van Antwerp, C. L., Eggert, H., Meakins, G. D., Miners, J. O., and Djerassi, C., Additivity relationships in carbon-13 nuclear magnetic resonance spectra of dihydroxy steroids.J. Org. Chem., 42, 789–793 (1977).
Wang, W., Li, R., Alam, N., Liu, Y., Lee, C.-O., Hong, J., Lee, C. K., Im, K. S., and Jung, J. H., New saponins from the starfishCertonardoa semiregularis.J. Nat. Prod., 65, 1649–1656 (2002).
Wang, W., Li, F., Park, Y., Hong, J., Lee, C.-O., Kong, J. Y., Shin, S., Im, K. S., and Jung, J. H., Bioactive sterols from the starfishCertonardoa semiregularis.J. Nat. Prod., 66, 384–391 (2003a).
Wang, W.,Li, F., Hong, J., Lee, C.-O., Cho, H. Y., Im, K. S.. and Jung, J. H., Four new saponins from the starfishCertonardoa semiregularis.Chem. Pharm. Bull., 51,435–439 (2003b).
Wang, W., Hong, J., Lee, C.-O., Im, K. S., and Jung, J. H., Cytotoxic sterols and saponins from the starfishCertonardoa semiregularis.J. Nat. Prod., 67,584–591 (2004a).
Wang, W., Hong, J., Lee, C.-O., Im, K. S., and Jung, J. H., Additional cytotoxic sterols and saponins from the starfishCertonardoa semiregularis.J. Nat. Prod., 67, 1654–1660 (2004b).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, W., Jang, H., Hong, J. et al. New cytotoxic sulfated saponins from the starfishCertonardoa semiregularis . Arch Pharm Res 28, 285–289 (2005). https://doi.org/10.1007/BF02977793
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02977793