Abstract
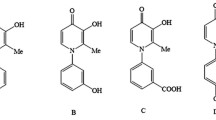
SK-302B, an antibiotic purified from soilStreptomyces sp. 302, was structurally identified as echinomycin (C50H66N11S2). In the present experiment, the possibility of SK-302B as an anticolon cancer agent was investigated by using chemosensitivity system (MTT assay, clonogenic assay). Treatment of SK-302B on various colon cancer cell lines resulted in a significant cytotoxicity and tumor colony formation inhibition. These studies showed that SK-302B had a potent inhibition on colon cancer cells.
Similar content being viewed by others
References Cited
Carmichael, J., deGraff, W.G., Gazdar, A.F., Minna, J.D. and Mitchel, J.B., Evaluation of a tetrazolium-based semiautomated colorimetric assay; assessment of radiosenstivity.Cancer. Res., 47, 936–942 (1987).
Geran, R. I., Greenberg, N.H., Macdonald, M.M., Schmacher, A.M. and Abott, B.J., Protocols for screeinig, chemical agents and natural products against animal tumors and other biological systems.Cancer. Chemother. Rep., part III 3, 1 (1972).
Greco, F.A., Richardson, R.L., Shulman and Oldmam, R.K., Combination of constant-infusion 5-fluorouracil, methyl-CCNU, mitomycin-C and vincristine in advanced colorectal carcinoma.Cancer. Treat. Rep., 62, 1407–1412 (1978).
Hutchison, C.R., Drug discovery and development through the genetic engineering of antibiotic-producing microorganisms.J. Med. Chem., 32, 936–937 (1989).
Kim, S.K. and Koh, C.M.,In vivo antitumor activity and toxicological study of SK-302B.J. Wonju Coll. Med., 6(1), 174 (1993).
Kim, S.K., Shin, W.S., Park, Y.S. and Choi, S.J.,In vitro chemosensitivity test fo SK-302B on human gastric carcinom, cell lines.J. Kor. Cancer. Ass., 27(5), 703–710 (1995).
Koh, C.M., Shin, W.S., Kim, S.K., Park, Y.S. and Park, J.Y., Antitumor activity of tumor cell growth inhibitory factor isolated fromStreptomyces sp.J. Kor. Soc. Microbiol., 29(3), 295–300 (1994).
Lee, N.K. and Jun, K.Y., A clinical study of colorectal cancer.J. Kor. Colo. Proctol. Soc., 6, 92–98 (1990).
Ministry of Health and Social Affairs, Five years report for cancer register program in the republic of Korea (1982.7.1–1989. 6.30).J. Kor. Cancer Ass., 21(1), 155–217 (1989).
Mitchell, M.S., Combining chemotherapy with biological response modifiers in treatment of cancer.J. Natl. Cancer Inst., 80, 1445–1450 (1989).
Salmon, S.E., Hamberger, A.W., Soehnlen, B., Durie, B.G.M., Alberts, D.S. and Moon, T.E., Quantitation of differential sensitivity of human tumor stem cells to anticancer drugs.N. Engl. J. Med., 298, 1321–1329 (1978).
Shoemaker, R.H., Igel, H.J., McLachlan, S.S. and Harfiel, J.L., Measurement of tumor colony growth and drug sensitivity in soft agarose culture using a ratio isotope method.Stem cells 1, 321–327 (1981).
Venditti, J.M., The National Cancer Institute antitumor drug discovery program. current and future perspectives: a commentary.Cancer. Treat Rep., 67, 767–772 (1983).
Von Hoff, D.D., Casper, J. and Bradely, E., Association between human tumor colony assay results and response of an individual patient's tumor to chemotherapy.Am. J. Med., 70, 1027–1032 (1981).
Wadler, S., Tenteromano, L., Cazenave, L., Sparano, J. A. and Greenwald, E.S., Phase II trial of echinomycin in patients with advanced or recurrent colorectal cancer.Cancer Chemother. Pharmacol., 34(3), 266–269 (1994).
Zubrod, C.G., The chemotherapy program of the National Cancer Institute: History, analysis and plans.Cancer Chemother. Rep., 50, 349–540 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, S.K., Ahn, C.M., Kim, TU. et al. In Vitro chemosensitivity test of SK-302B on human colon carcinoma cell lines. Arch. Pharm. Res. 19, 261–263 (1996). https://doi.org/10.1007/BF02976237
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02976237