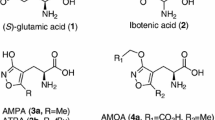
Abstract
Bioisostere approach has been shown to be useful to augment potency or to modify certain physiological properties of a lead compound. Based upon well documented bioisosterism, an isosteric replacement of benzene ring of 4-hydroxy-2-quinolone compound (L-695902) with a thiophene moiety was carried out to prepare the title compounds, 4-hydroxy-6-oxo-6,7-dihydro-thieno[2,3-b] pyrimidines15. The resulting bioisosteric compounds15 were evaluated for their antagonistic activity (binding assay) for NMDA receptor glycine site.
Similar content being viewed by others
References
Blair, J. B., Kanthasamy, A., Lucaites, V. L., Nelson, D. L., and Nichols, D. E., Thieno[3,2-b]-and Thieno[2,3-b] pyrrole Bioisosteric Analogues of the Hallucinogen and Serotonin AgonistN, N-Dimethyltryptamine.J. Med. Chem. 42, 1106–1111 (1999).
Cai, S. X., Kher, S. M., Zhou, Z. L., Ilyin, V., Espitia, S. A., Tran, M., Hawkinson, S. E., Woodward, R. M., Weber, E., and Keana, J. F., Structure-activity relationships of alkyl-and alkoxy-substituted 1,4-dihydroquinoxaline-2, 3-diones: Potent and systemically active antagonists for the glycine site of the NMDA receptor.J. Med Chem., 40, 730–738 (1997).
Epperson, J. R., Hewawasam, P., Meanwell, N. A., Boissard, C. G., Cribkoff, V. K., and Postmunson, D., Synthesis and excitatory Amino acid pharmacology of some novel quinoxalinediones.Biomed. Chem. Lett., 3, 2801–2804 (1993).
Fabio, R. D., Capelli, A. M., Conti, N., Cugola, A., Donati, D., Feriani, A., Gastaldi, P., and Gaviraghi, G., Substituted indole-2-carboxylates as in vivo potent antagonists acting as the strychnine-insensitive glycine binding site.J. Med. Chem., 40, 841–850 (1997).
Honore, T., Davies, S. N., Drejer, J., Fletcher, E. J., Jacobsen, P., Lodge, D., and Nielsen, F. E., Quinoxalinediones: Potent competitive non-NMDA glutamate receptor antagonists.Science, 241, 701–703 (1988).
Hwang, K. J., and Choi, N. K., A Facile Synthesis of 2-Aminothiophene Derivatives.Bull. Korean. Chem. Soc. 12, 121–122 (1991).
Iversen, L. L. and Kemp, J. A., Noncompetitive NMDA antagonists as drugs. In The NMDA Receptor, 2nd ed., Collingridge, G. L., Watkins, J. C. (Eds.), IRL press, Oxford, England, pp 469–486 (1994).
Johnson, J. W. and Ascher, P., Glycine potentiates the NMDA response in cultured mouse brain neurons.Nature, 325, 529–531 (1987).
Johnson, R. L. and Koerner, K. F. Excitatory amino acid Neurotransmission.,J. Med. Chem. 31, 2057–2066 (1988).
Kemp, J. A. and Leeson, P. D. The glycine site of the NMDA receptor.Trends pharmacol. Sci., 14, 20–25 (1993).
Kulagowski, J. J., Baker, R., Cirtis, N. R., Leeson, P. D., Mawer, I. M., Moseley, A. M., Ridgill, M. P., Rowley, M., Stansfield, I., Foster, A. C., Gromwood, S., Hill, R. G., Kemp, J. A., Marshall, G. R., and Saywell, K. L., Tricklebank, M. D. 3′-(Arylmethyl- and 3′-Aryloxy)-3-phenyl-4-hydroxyquinolin-2(1 H)-ones: Orally Active Antagonists of the Glycine Site on the NMDA Receptor.J. Med. Chem. 37, 1402–1405 (1994).
Kulagowski, J. J., Glycine-site NMDA receptor antagonists:An update, Exp. Opin. Ther. Pat. 6, 1069–1079 (1996).
Leeson, P. D., Baker, R., Carling, R. W., Curtis, N. R., Moore, K. W., William, B. J., Foster, A. C., Donald, A. E., Kemp, J. A., and Marshall, G. R., Kynurenic acid derivatives. Structure-activity relationships for excitatory amino acid antagonism and identification of potent and selective antagonists at the glycine site on the NMDA receptor,J. Med. Chem., 34, 1243–1252 (1991).
Leeson, P. D., Carling, R. W., Moore, K. W., Moseley, A. M., Smith, G. D., Stevenson, G., Chan, T., Baker, R., Foster, A. C., Grimwood, S., Kemp, G. A., Marshall, G. R., and Hoogsteen, K., 4-Amido-2-carboxytetrahydro-quinolines. Structure-activity relationships for antagonism at the glycine site of the NMDA receptor,J. Med. Chem., 35, 1954–1968 (1992).
Leeson, P. D. and Iversen, L. L., The glycine site on the NMDA receptor: Structure-activity relationship and therapeutic potential,J. Med. Chem., 37, 4053–4067, (1994).
McQuaid, L. A., Smith, E. C. R., Lodge, D., Pralong, E., Wikel, J. H., and Calligaro, D. O., 3-Phenyl-4-hydroxy-2-quinolin-2(1H)-ones: Potent and Selective antagonists at the strychnine-insensitive glycine site on the NMDA receptor complex,J. Med. Chem., 35, 3423–3425 (1992).
McCullough, J., Excitatory amino acids antagonists and their potential for the treatment of ischaemic brain damage in man.J. Gin. Parmaco., 34, 106–114 (1992).
Meldrum, B., Protection against ischaemic neuronal damage by drugs acting on excitatory neurotransmission.Cerebrovasc. Brain Metab. Rev., 2, 27–57 (1990).
Monagham, D. T., Bridges, R. J., and Cotman, C. W., The Excitatory Amine acid Receptor; Their Classes, Pharmacology and Distinct properties in the Function of the Central Nerveous System.Annu. Rev. Phamacol. Toxicol. 29, 365–402 (1989).
Rothman, S. M. and Olney, J. W., Excitotoxicity and the NMDA receptor,Trends Neurosci., 10, 299–302 (1987).
Rowley, M., Leeso, P. D., Stevenson, G. I., Moseley, A. M., Stansfield, I., Sanderson, I., Robinson, L., Baker, R., Kemp, J. A., Marshall, G. R., Foster, A. C., Grimwood, S., Tricklebank, M. D., and Saywell, K. L., 3-Acyl-4-hydroxy-quinolin-2(1H)-ones. Systemically Active Anticonvulsants Acting by Antagonism at the Glycine Site of the N-Methyl-D-aspartate Receptor Complex.J. Med. Chem. 36, 3386–3396 (1993).
Salituro, F. G., Harrison, B. L., Baron, B. M., Nyce, P. L., Stewart, K. T., Kehne, J. H., White, H. S., and McDonald, I. A., 3-(2-carboxyindol-3-yl)propionic acid-based antagonists of the NMDA receptor associated glycine binding site.,J. Med. Chem., 35, 1791–1799 (1992).
Silverman, R. B., The Organic Chemistry of Drug Design and Drug Action. Academic Press, New York, pp. 19–24 (1992).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hwang, KJ., Lee, TS., Kim, KW. et al. 4-Hydroxy-6-Oxo-6,7-Dihydro-Thieno[2,3-b] pyrimidine derivatives: Synthesis and their biological evaluation for the glycine site acting on theN-Methyl-D-aspartate (NMDA) receptor. Arch Pharm Res 24, 270–275 (2001). https://doi.org/10.1007/BF02975090
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02975090