Abstract
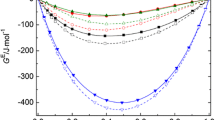
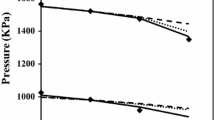
Data on the solubility ofp-hydroxybenzoate in amide-water cosolvent system and surface tension of the binary amide-water cosolvents are analyzed in terms of the possible mechanism for cosolvency. The results of the study suggest that strong partitioning of the alkylated amides to the ester surface, thus reducing the hydrophobic interface within the system, may account for much of the cosolvency phenomena observed in these systems.
Similar content being viewed by others
Literature Cited
Yalkowsky, S. H., Amidon, G. L., Zografi, G. and Flynn, G. L., Solubility of nonelectrolytes in polar solvents(III), Alkylp-aminobenzoates in polar and mixed solvents.J. Pharm. Sci. 64, 48(1975).
Lindstrom, R. E.,J. Pharm. Sci. (in press).
Lee, C. H., and Lindstrom, R. E., Solubility in amide-water cosolvent system(III), A thermodynamic view.J. Pharm. Sci. (in press).
Lee, C. H., and Lindstrom, R. E., Solubility in amide-water cosolvent(IV), Amide adsorption to the air/water interface.Drug Devel. Ind. Pharm. (in press).
Giaquinto, A. R., Lindstrom, R.E., Swarbrick J., and Losurdo, A.,J. Solution Chem. 6, 687(1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, C.H., Lindstrom, R.E. Interfacial and free energy effects in aqueous amide cosolvents. Arch. Pharm. Res. 2, 85–88 (1979). https://doi.org/10.1007/BF02959034
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02959034