Abstract
The major class of mycotoxins produced byFusarium moulds are trichothecenes, a large group of sesquiterpenes sharing the same basic chemical structure, a 12,13-epoxytrichothec-9-ene ring system. Their toxicity is attributed to their ability to noncompetitively inhibit the biosynthesis of proteins in eukaryotic cells. Trichothecenes in general are relatively stable substances and their degradation is reported only at high temperatures and prolonged heating time.
In an attempt to investigate the stability of the trichothecene nivalenol (NIV) under food processing conditions such as cooking or baking, we performed a number of experiments using a model heating system.
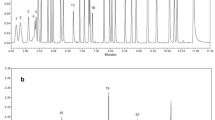
Heating of nivalenol, especially under mild alkaline conditions, gave a mixture of four compounds (norNIV A, norNIV B, norNIV C and NIV lactone), which were separated and further analyzed by gas chromatography/mass spectrometry (GC/MS) and liquid chromatography/mass spectrometry (LC/MS). Structure elucidation was achieved by 1D and 2D nuclear magnetic resonance (NMR) experiments.
We further demonstrated the formation of these products in heating experiments with spiked flour samples. In a screening of several commercially available products only norNIV B was detected in one of the samples, possibly due to the very low contamination of these particular samples with nivalenol.
Similar content being viewed by others
Literatur
European Commission, Scientific Committee On Food (2002) Opinion Of The Scientific Committee On Food On Fusarium Toxins, Part 6: Group evaluation of T-2 toxin HT-2 toxin, nivalenol and deoxynivalenol
European Commission, Scientific Committee On Food (2000) Opinion Of The Scientific Committee On Food On Fusarium Toxins, Part 4: Nivalenol
Lauren DR, Smith WA (2001) Stability of theFusarium mycotoxins nivalenol, deoxynivalenol and zearalenone in ground maize under typical cooking environments. Food Additives and Contaminants 18 (11): 1011–1016
Wolf C, Bullerman L (1997) Heat and pH alter the concentration of deoxynivalenol in an aqueous environment. Journal of Food Protection 61 (3): 365–36
Abbas H, Mirocha C, Rosiles R, Carvajal M (1988) Decomposition of Zearalenone and Deoxynivalenol in the process of making tortillas from corn. Cereal Chemistry 65 (1): 15–19
Wolff J (2004) Untersuchungen der Gehaltsveränderungen der Fusariumtoxine Deoxynivalenol und Zearalenon durch Be-und Verarbeitungsprozesse in Getreide und Getreideprodukten, Vortrag auf dem 26. Mykotoxin-Workshop, Herrsching, Germany.
Seefelder W, Knecht A, Humpf H-U (2003) Bound fumonisin B1: Analysis of fumonisin-B1 glyco and amino acid conjugates by liquid chromatography-electrospray ionization-tandem mass spectrometry. J. Agric. Food Chem. 51: 5567–5573
Young JC, Blackwell BA, Apsimon JW (1986) Alkaline Degradation Of The Mycotoxin 4-Deoxynivalenol. Tetrahedron Letters 27: 1019–1022
Grove JF (1985) Phytotoxic compounds produced byFusarium equiseti. Part 7: Reactions and Rearrangement of the 7-Hydroxy-12, 13-epoxytrichothec-9-en-8-one skeleton. J. Chem. Soc. Perkins Trans. 1: 1731–1736
Bretz M, Knecht A, Göckler S, Humpf H-U (2005) Structural elucidation and analysis of thermal degradation products of theFusarium mycotoxin nivalenol. Mol. Nutr. Food research, im Druck
Smith M, Lewis C, Anderson J, Solomons G (1994) Mycotoxins in human nutrition and health, Directorate-General XII, Science, Research and Development, European Commission
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bretz, M., Göckler, S. & Humpf, H.U. Isolierung und Strukturaufklärung von thermischen Abbauprodukten des Mykotoxins Nivalenol. Mycotox Res 21, 15–17 (2005). https://doi.org/10.1007/BF02954807
Issue Date:
DOI: https://doi.org/10.1007/BF02954807