Abstract
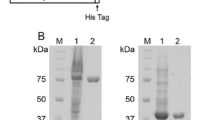
A chitinase gene (pCHi58) encoding a 58 kDa chitinase was isolated from theSerratia marcescens KCTC 2172 cosmid library. The chitinase gene consisted of a 1686 bp open reading frame that encoded 562 amino acids.Escherichia coil harboring the pChi58 gene secreted a 58 kDa chitinase into the culture supernatant. The 58 kDa chitinase was purified using a chitin affinity column and mono-S column. A nucleotide andN-terminal amino acid sequence analysis showed that the 58 kDa chitinase had a leader peptide consisting of 23 amino acids which was cleaved prior to the 24th alanine. The 58 KDa chitinase exhibited a 98% similarity to that ofS. marcescens QMB 1466 in its nuclotide sequence. The chitinolytic patterns of the 58 kDa chitinase released N,N′-diacetyl chitobiose (NAG2) as the major hydrolysis end-product with a trace amount ofN-acetylglucosamine. When a 4-methylumbellyferyl-N-acetylglucosamin monomer, dimmer, and tetramer were used as substrates, the 58 kDa chitinase did not digest the 4-Mu-NAG monomer (analogue of NAG2), thereby indicating that the 58 kDa chitinase was likely an endochitinase. The optimum reaction temperature and pH of the enzyme were 50°C and 5.0, respectively.
Similar content being viewed by others
References
Gooday, G. W., A. M. Humphreys, and W. H. McIntosh. (1986) Roles of chitinases in fungal growth In: R. Muzzarelli, C. Jeuniaux, and G. W. Gooday, (eds.).Chitin in Nature and Technology, pp. 83–91. Plenum Press, New York, USA.
Chtakara, A. (1961) Studies on the chitinolytic enzymes of black-koji mold: Purification of chitinase.Agric. Biol. Chem. 25: 54–60.
Boller, T. (1985) Induction of hydrolase as a defense reaction against pathogen, In: J. L. Key and T. Kosuge (eds.),Cellular and Molecular Biology of Plant Stress. pp. 247–262, A. R. Liss, New York, USA.
Powning, R. F. and H. Irzykiewez (1965) Studies on chitinase systems in bean and other seeds.Comp. Biochem. Physiol. 14: 127–133.
Hamamura, Y. and Y. Kanehara (1940) Enzymatyic studies on exuvial fluid ofBombyx mori: Chitinase.J. Agric. Chem. Soc. Jpn. 16: 907–909.
Lunt, M. R. and P. W. Kent (1960) Chitinase system fromCarcinus maenas.Biochem Biophys. Acta 44: 371–373.
Clarke, P. H. and M. V. Tracey (1956) The occurrence of chitinase in some bacteria.J. Gen. Microbiol. 14: 188–196.
Roberts, R. L. and E. Cabib (1982)Serratia marcescens chitinase: one-step purification and use for determination of chitin.Anal. Biochem. 127: 402–412.
Watanabe, T., K. Suzuki, W. Oyanagi, K. Ohnishi, and H. Tanaka (1990) Gene cloning of chitinase A1 fromBacillus circulans WL-12 revealed its evolutionary relationship toSerratia chitinase and to the type III homology units of fibronectin.J. Biol. Chem. 265: 15659–15665.
Shinshi, H., D. Mohnen, and F. Meins (1987) Regulation of a plant pathogenesis-related enzyme: inhibition of chitinase and chitinase mRNA accumulation in cultured tabacco tissues by auxin and cytokinin.Proc. Natl. Acad. Sci. USA 84: 89–93.
Fuchs, R. L., S. A. Mcpherson, and D. J. Drahos (1986) Cloning of aSerratia marcescens gene encoding chitinase.Appl. Environ. Microbiol. 51: 504–509.
Tsujibo, H., H. Orikoshi, K. Fujimoto, K. Miyamoto, C. Imada, Y. Okami, and Y. Inamori (1993) Cloning, sequence, and expression of a chitinase gene from a marine bacterium,Altermonas sp strain O-7.J. Bacteriol. 175: 178–181.
Wortman, A. T., C. C. Somerville, and R. R. Colwell (1986) Chitinase determinants ofVibrio vulnificus: Gene cloning and applications of a chitinase probe.Appl. Environ. Microbiol. 52: 142–145.
Sambrook, J., E. F. Fritsch, and T. Maniatis (1989)Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, USA.
Gal, S. W., J. Y. Choi, C. Y. Kim, Y. H. Cheong, Y. J. Choi, J. D. Bahk, S. Y. Lee, and M. J. Cho (1997) Isolation and characterization of the 54 kDa and 22 kDa chitinase genes ofSerratia marcescens KCTC 2172.FEMS Microbiol. Lett. 151: 197–204.
Gal, S. W., J. Y. Choi, C. Y. Kim, Y. H. Cheong, Y. J. Choi, J. D. Bahk, S. Y. Lee, and M. J. Cho (1998) Cloning of the 52-kDa chitinase gene fromSerratia marcescens KCTC 2172 and its proteolytic cleavage into an active 35-kDa enzyme.FEMS Microbiol. Lett. 160: 151–158.
Huber, M., E. Cabib, and L. H. Miller (1991)Proc. Natl. Acad. Sci. USA 88: 2807–2810.
Vinetz, J. M., J. G. Valenzuela, C. A. Specht, L Aravind, R. C. Langer, J. M. C. Ribeiro, and D. C. Kaslow (2000) Chitinases of the Avian Malaria parasitePlasmodium gallinaceum, a class of enzymes necessary for parasite invasion of the mosquito midgut.J. Biol. Chem. 275: 10331–10341.
Kless, H., Y. Sitrit, I. Chet, and A. B. Oppenheim (1989) Cloning of the gene coding for chitobias ofSerratia marcescens.Mol. Gen. Genet. 217: 471–473.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gal, S.W., Lee, S.W. & Choi, Y.J. Molecular cloning and characterization of 58 kDa chitinase gene fromSerratia marcescens KCTC 2172. Biotechnol. Bioprocess Eng. 7, 38–42 (2002). https://doi.org/10.1007/BF02935878
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02935878