Abstract
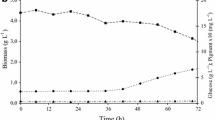
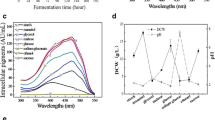
To maximize and sustain the productivity ofMonascus pigments, various environmental and nutritional parameters, such as the initial moisture content, pH, inoculum size, sample size, and nutrient supplement, that influence pigment production were evaluated in solid-state cultures as follows: initial moisture content, 50%; pH, 6.0; inoculum size 1 x 104 spore cells (grams of dry solid substrate)−1; sample size, 300 g. All supplementary nutrients (carbon, nitrogen, and mineral sources) added has inhibitory effects on the cell growth and red pigment production. In open tray culture the maximum biomass yield and specific productivity of red pigments were 223 mg DCW (grams of initial dry substrate)−1 and, 47.6 OD500 (DCW grams)−1 h−1, respectively.
Similar content being viewed by others
References
Lin, C. F. (1973) Isolation and culture condition ofMonascus sp. for the production of pigment in submerged culture.J. Ferment. Technol. 51: 407–414.
Pastrana, L., P. J. Blanc, M. O. Santerre, O. Loret, and G. Goma (1995) Production of red pigments byMonascus ruber in synthetic media with a strictly controlled nitrogen source.Process Biochem. 30: 333–341.
Chen, M. H. and M. R. Johns (1993) Effect of pH and nitrogen source on pigment production byMonascus purpureus.Appl. Microbiol. Biotechnol. 40: 132–138.
Carels, M. and D. Shepherd (1977) The effect of different nitrogen sources on pigment production and sporulation ofMonascus species in submerged shaken culture.Can. J. Microbiol. 23: 1360–1372.
Mchan, F. and G. T. Johnson (1979) some effects of zinc on the utilization of nitrogen sources byMonascus purpureus.Mycologia 71: 160–169.
Broder, C. U. and P. E. Koelher (1980) Pigment produced byMonascus purpureus with regard to quality and quantity.J. Food Sci. 45: 567–569.
Lee, Y.-K., D.-C. Chen, S. Chauvatcharin, T. Seki, and T. Yoshida (1995) Production ofMonascus pigments by a solid-liquid state culture method.J. Ferment. Bioeng. 79: 516–518.
Mulimani, V. H., G. N. Patil, and Ramalingam (2000) Amylase production by solid-state fermentation: A new practical approach to biotechnology courses.Biochemical Education 28: 161–163.
Juzlova, P., I. Martincova, and J. Lozinski (1994) Eshanol as substrate for pigment production by the fungusMonascus.Enzyme Microbiol. Technol. 16: 996–1001.
Adidoo, K. E., R. Hendry, and B. J. B. Wood (1981) Estimation of fungal growth in a solid state fermentation system.Appl Microbiol. Biotechnol. 12: 6–9.
Lotong, N. and P. Suwanarit (1990) Fermentation of angkak in plastic bags and regulation of pigment by initial moisture content.J. Appl. Bacteriol. 68: 565–570.
Ross, I. S. (1975) Some effects of heavy metals on fungal cells.Trans. Br. Mycol. Soc. 64: 175–795.
Bau, Y. S. and H. C. Wong (1979) Zinc effects on growth pigmentation and antibacterial activity ofMonascus purpureous.Physiol. Plant. 46: 63–67.
Zhang, M., H. Chang, and C. Lin (1982) Studies on the red pigment production ofMonascus: Strain and fermentation.Shipin Yu Faxiao Gongyu. 4: 1–7.
Mudgett, R. E. and A. J. Paradis (1985) Solid-state Fermentation of natural brinch lignin byPhanerochaete Chrysosporium.Enzyme Microb Technol. 7: 150–154
Turnet, W. B. (1971)Fungal Metabolites. 2nd ed., Academic Press, London, UK.
Han, S. and R. E. Mudgen (1992) Effects of oxygen and carbon dioxide partial pressures onMonascus growth and pigment production in solid-state fermentations.Biotechnol. Prog. 8: 5–10.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, BK., Piao, H.Y. & Chung, WJ. Production of red pigments byMonascus purpureus in solid-state culture. Biotechnol. Bioprocess Eng. 7, 21–25 (2002). https://doi.org/10.1007/BF02935875
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02935875