Abstract
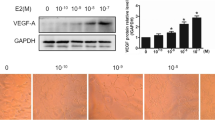
Although 17β-estradiol (E2) is known to induce upregulation of endothelial nitric oxide synthase (eNOS) gene expression, the underlying mechanism is largely unclear. In this study, we show that 17β-estradiol (E2) increases eNOS expression through the binding of estrogen receptor α (ERα) to its promoter. E2 (1 nM) increased both eNOS and nitric oxide production, peaking at 24 h, in bovine aortic endothelial cells, and these increases were accompanied by increment of ERα. To map the estrogen-responsive elements of the eNOS gene for ERα binding under E2, we used three constructs, which were an eNOS gene promoter (−1,600 to +22 nucleotides) fused with a luciferase reporter gene (pGL2-eNOS(−1,600)). However, progressive 5′-deletion from −1,600 to −429 of promoter was attenuated the eNOS transcription. Electrophoretic mobility shift and anti-ERα antibody supershift analyses showed that ERα bound to the estrogen-responsive element (ERE) site, located at −756 to −748 of the eNOS promoter. Our data demonstrate that the interaction between ERα and the estrogenresponsive elements within the eNOS promoter may represent a mechanism for E2-induced gene expression.
Similar content being viewed by others
References
Giselard, V., V. M. Miller, and P. M. Vanhoutte (1988) Effect of 17 beta-estradiol on endothelium-dependent responses in the rabbit.J. Pharmacol. Exp. Ther. 244: 19–22.
Noguchi, S., M. Nakatsuka, K. Asagiri, T. Habara, M. Takata, H. Konishi, and T. Kudo (2002) Bisphenol A stimulates NO synthesis through a non-genomic estrogen receptor-mediated mechanism in mouse endothelial cells.Toxicol. Lett. 135: 95–101.
Venkov, C. D., A. B. Rankin, and D. E. Vaughan (1996) Identification of authentic estrogen receptor in cultured endothelial cells: A potential mechanism for steroid hormone regulation of endothelial function.Circulation 94: 727–733.
Tan, E., M. V. Gurjar, R. V. Sharma, and R. C. Bhalla (1999) Estrogen receptor-α gene transfer into bovine aortic endothelial cells induces eNOS gene expression and inhibits cell migration.Cardiovasc. Res. 43: 788–797.
Isozumi, K., Y. Fukuuchi, H. Takeda, and Y. Itoh (1994) Mechanisms of CBF augmentation during hypoxia in cats: probable participation of prostacyclin, nitric oxide and adenosine.Keio J. Med. 43: 31–36.
Karas, R. H., B. L. Patterson, and M. E. Mendelsohn (1994) Human vascular smooth muscle cells contain functional estrogen receptor.Circulation 89: 1943–1950.
Greenberg, B. and S. Kishiyama (1993) Endothelium-dependent and- independent responses to severe hypoxia in rat pulmonary artery.Am. J. Physiol. 265: H1712-H1720.
Leach, R. M., H. S. Hill, V. A. Snetkov, and J. P. Ward (2002) Hypoxia, every state and pulmonary vasomotor tone.Respir. Physiol. Neurobiol. 132: 55–67.
Sumi, D. and L. J. Ignarro (2003) Estrogen-related receptor α1 up-regulates endothelial nitric oxide synthase expression.Proc. Natl. Acad. Sci. USA 100: 14451–14456.
Kim, H. P., J. Y. Lee, J. K. Jeong, S. W. Bae, H. K. Lee, and I. Jo (1999) Nongenomic stimulation of nitric oxide release by estrogen is mediated by estrogen receptor α localized in caveolae.Biochem. Biophys. Res. Commun. 263: 257–262.
Cho, D.-H., Y. J. Choi, S. A. Jo, and I. Jo (2004) Nitrie oxide production and regulation of endothelial nitric oxide synthase phosphorylation by prolonged treatment with troglitazone: Evidence for involvement of PPAR gamma-dependent and PPARgamma-independent signaling pathways.J. Biol. Chem. 279: 2499–2506.
Lee, K. A., A. Bindereif, and M. R. Green (1988) A small-scale procedure for preparation of nuclear extracts that support efficient transcription and pre-mRNA splicing.Gene Anal. Tech. 5: 22–31.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Min, J. 17β-estradiol-stimulated eNOS gene transcriptional activation is regulated through the estrogen-responsive element in eNOS promoter. Biotechnol. Bioprocess Eng. 12, 446–449 (2007). https://doi.org/10.1007/BF02931069
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02931069