Abstract
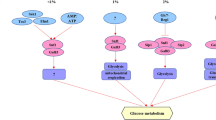
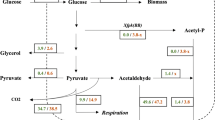
Since Gcr1p is pivotal in controlling the transcription of glycolytic enzymes and trehalose metabolism seems to be one of the control points of glycolysis, we examined trehalose and glycogen synthesis in response to 2 % glucose pulse during batch growth ingcr1 (glucose regulation-1) mutant lacking fully functional glycolytic pathway and in the wild-type strain. An increase in both trehalose and glycogen stores was observed 1 and 2 h after the pulse followed by a steady decrease in both the wild-type and thegcr1 mutant. The accumulation was faster while the following degradation was slower ingcr1 cells compared to wild-type ones. Although there was no distinct glucose consumption in the mutant cells it seemed that the glucose repression mechanism is similar ingcr1 mutant and in wild-type strain at least with respect to trehalose and glycogen metabolism.
Similar content being viewed by others
Abbreviations
- Gle:
-
glucose
- Glg:
-
glycogen
- Tre:
-
trehalose
- UAS:
-
upstream activator sequence
- YP:
-
yeast extract-peptone
- YPG:
-
yeast extract-peptone-glycerol
- YPGal:
-
yeast extract-peptone-galactose
- YPGL:
-
yeast extract-peptone-glycerol-lactate
References
Bell W., Weining S., Hohmann S., Wera S., Reinders A., De Virgillio C., Wiemken A., Thevelein J.M.: Compositional and functional analysis of theSaccharomyces cerevisiae trehalose synthase complex.J.Biol.Chem.273, 33311–33319 (1998).
Boles E., Zimmermann F.K.: Induction of pyruvate decarboxylase in glycolysis mutants ofS. cerevisiae correlates with the concentration of three-carbon glycolytic intermediates.Arch.Microbiol.160, 324–328 (1993).
Boles E.: Pyruvate kinase, pp. 171–186 in F.K Zimmermann, K.D. Entian (Eds):Yeast Sugar Metabolism: Biochemistry, Genetics, Biotechnology and Applications. Technomics, Lancaster (USA) 1997.
Boles E., Schulte F., Miosga T., Freidel K., Schluter E., Zimmermann F.K., Hoolenberg C., Heinisch J.: Characterization of a glucose repressed pyruvate kinase (Pyk2) inSaccharomyces cerevisiae that is catalytically insensitive to fructose 1,6-bisphosphate.J.Bacteriol.179, 2987–2993 (1997).
Clifton D., Fraenkel D.G.: Thegcr (glycolysis regulation) mutation ofS. cerevisiae.J.Biol.Chem.256, 13074–13078 (1981).
Denis C.L., Ferguson J., Young E.: mRNA levels for the fermentative alcohol dehydrogenase ofSaccharomyces cerevisiae decrease upon growth on a nonfermentable carbon source.J.Biol.Chem.258, 1165–1171 (1983).
François J., Villanueva M.E., Hers H.G.: The control of glycogen metabolism in yeast — 1. Interconversionin vivo of glycogen synthase and glycogen phosphorylase induced by glucose, a nitrogen source or uncouplers.Eur.J.Biochem.174, 551–559 (1988).
François J.M., Blazquez M.A., Arino J., Gancedo C.: Storage carbohydrates in the yeastSaccharomyces cerevisiae, pp. 287–311 in F.K. Zimmermann, K.D. Entian (Eds):Yeast Sugar Metabolism: Biochemistry, Genetics, Biotechnology and Applications. Technomics, Lancaster (USA) 1997.
François J., Parrou J.L.: Reserve carbohydrates metabolism in the yeastSaccharomyces cerevistae.FEMS Microbiol.Rev.25, 125–145 (2001).
Herrero P., Flores L., De la Cera T., Moreno F.: Functional characterization of transcriptional regulatory elements in the upstream region of the yeastGLK1 gene.Biochem.J.343, 319–325 (1999).
Hohmann S., Neves M.J., de Koning W., Alijo R., Ramos J., Thevelein J.M.: The growth and signaling defects of theggs1 (fdp1/byp1) deletion mutant on glucose are suppressed by a deletion of the gene encoding hexokinase PII.Curr.Genet.23, 281–289 (1993).
Hottiger T., Boller T., Wiemken A.: Rapid changes of heat and desiccation tolerance correlated with changes of trehalose content inSaccharomyces cerevisiae cells subjected to temperature shifts.FEBS Lett.220, 113–115 (1987).
Johnston M.: Feasting, fasting and fermenting.Trends Genet.15, 29–33 (1999).
Lillie S.H., Pringle J.R.: Reserve carbohydrate metabolism inSaccharomyces cerevisiae: response to nutrient limitation.J.Bacteriol.143, 1384–1394 (1980).
Londesborough J., Vuorio O.: Purification of trehalose synthase from bakers’ yeast — its temperature dependent activation by fructose 6-phosphate and inhibition by phosphate.Eur.J.Biochem.216, 841–848 (1993).
Lopez M., Baker V.H.: Understanding the growth phenotype of the yeastger1 mutant in terms of global genomic expression patterns.J.Bacteriol.182, 4970–4978 (2000).
Ma P., Goncalves T., Matetzek A., Dias M.C.L., Thevelein J.: The lag-phase rather than the exponential growth phase on glucose is associated with a higher cAMP level in wild type and cAPK-attenuated strains of the yeastSaccharomyces cerevisiae.Microbiology143, 3451–3459 (1997).
Meijer M.C.M., Boonstra J., Verkieli A.R., Verrips C.T.: Glucose repression inSaccharomyces cerevisiae is related to the glucose concentration rather than the glucose flux.J.Biol.Chem.273, 24102–24107 (1998).
Parrou J.L., François J.: A simplified procedure for a rapid and reliable assay of both glycogen and trehalose in whole yeast cells.Anal.Biochem.248, 186–188 (1997).
Pernambuco M.B., Windericks J., Crauvels M., Grifeon G., Mager M.H., Thevelein J.M.: Glucose triggered signaling inS. cerevisiae: different requirements for sugar phosphorylation between cells grown on glucose and those grown on nonfermentable carbon sources.Microbiology142, 1775–1782 (1996).
Santangelo M., Tornow J.: Efficient transcription of the glycolytic geneADH1 and three translational components requires theGCR1 product, which can act throughTUF/GRF1/RAP binding sites 1.Mol.Cell.Biol.10, 859–862 (1990).
Scott E.W., Allison H.E., Baker H.: Characterization ofTP1 gene expression in isogenic wild-type andgcrl-delection mutant strains ofSaccharomyces cerevisiae.Nucl.Acids Res.18, 7099–7107 (1990).
Steward P.R.: Analytical methods for yeasts, pp. 11–145 in D.W. Prescot (Ed.):Methods in Cellular Biology, Vol. 10. Academic Press, New York 1975.
Thevelein J.M.: Signal transduction in yeast.Yeast10, 1753–1790 (1994).
Thevelein J.M., Hohmann S.: Trehalose synthase: guard to the gate of glycolysis in yeast?Trends Biol.Sci.20, 3–10 (1995).
Türkel S.: TheGCR1 gene function is essential for glycogen and trehalose metabolism inSaccharomyces cerevisiae.Folia Microbiol.47, 663–666 (2002).
Uemura H., Fraenkel D.G.:gcr2, a new mutation affecting glycolytic gene expression inS. cerevisiae.Mol.Cell.Biol.10, 6389–6396 (1990).
Uemura H., Fraenkel D.G.: Glucose metabolism ingcr mutants ofSaccharomyces cerevisiae.J.Bacteriol.181, 4719–4723 (1999).
Uemura H., Jigami Y.: Role ofGCR2 in transcriptional activation of yeast glycolytic genes.Mol.Cell.Biol.12, 3834–3842 (1992).
Uemura H., Koshi M., Inoue Y., Lopez C., Baker H.: The role of Gcr1p in the transcriptional activation of glycolysis genes in yeastS. cerevisiae.Genetics147, 521–532 (1997).
Van Dijck P., Colavizza D., Smet P., Thevelein T.M.: Differential importance of trehalose in stress resistance in fermenting and nonfermentingSaccharomyces cerevisiae cells.Appl.Environ.Microbiol.61, 109–115 (1995).
Van Leare A.: Trehalose, reserve and/or stress metabolite.FEMS Microbiol.Rev.63, 201–210 (1989).
Wiemken A.: Trehalose in yeast, stress protectant rather than reserve carbohydrate.Antonie van Leeuwenhoek58, 209–217 (1990).
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by grant no. TOCTAG-2730 of theScientific and Technical Research Council of the Turkish Republic (TUBITAK) and by grant no. AFP-2000-07-02-12 of theInstitute of Natural and Applied Science. Middle East Technical University (Ankara).
Rights and permissions
About this article
Cite this article
Şeker, T., Hamamci, H. Trehalose, glycogen and ethanol metabolism in thegcrl mutant ofSaccharomyces cerevisiae . Folia Microbiol 48, 193–198 (2003). https://doi.org/10.1007/BF02930955
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02930955