Abstract
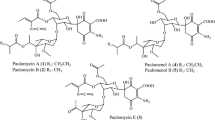
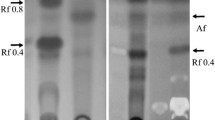
The ability to transform biologically exogenous daunomycinone, 13-dihydrodaunomycinone, aklavinone, 7-deoxyaklavinone, ε-rhodomycinone, ε-isorhodomycinone and ε-pyrromycinone was studied in submerged cultures of the following strains: wildStreptomyces coeruleorubidus JA 10092 (W1) and its improved variants 39–146 and 84–17 (type P1) producing glycosides of daunomycinone and of 13-dihydro-daunomycinone, together with ε-rhodomycinone, 13-dihydrodaunomycinone and 7-deoxy-13-dihydro-daunomycinone; in five mutant types ofS. coeruleorubidus (A, B, C, D, E) blocked in the biosynthesis of glycosides and differing in the production of free anthracyclinones; in the wildStreptomyces galilaeus JA 3043 (W2) and its improved variant G-167 (P2) producing glycosides of ε-pyrromycinone and of aklavinone together with 7-deoxy and bisanhydro derivatives of both aglycones; in two mutant typesS. galilaeus (F and G) blocked in biosynthesis of glycosides and differing in the occurrence of anthracyclinones. The following bioconversions were observed: daunomycinone → 13-dihydrodaunomycinone and 7-deoxy-13-di-hydrodaunomycinone (all strains); 13-dihydrodaunomycinone → 7-deoxy-13-dihydrodaunomycinone (all strains); daunomyeinone or 13-dihydrodaunomycinone → glycosides of daunomyeinone and of 13-dihydrodaunomycinone, identical with metabolites W1 and P1 (type A), or only a single glycoside of daunomyeinone (type E); aklavinone → ε-rhodomycinone (types A and B); aklavinone → 7-deoxyaklavinone and bisan-hydroaklavinone (type C); ε-rhodomycinone → ζ-rhodomycinone (types C, E); ε-rhodomycinone → glycosides of ε-rhodomycinone (types W2, P2); ε-isorhodomycinone → glycosides of ε-isorhodomycinone (types W2, P2); ε-pyrromycinone → a glycoside of ε-pyrromycinone (types W1, P1). 7-Deoxyaklavinone remained intact in all tests. Exogenous daunomyeinone suppressed the biosynthesis of its own glycosidea in W1 and P1; it simultaneously increased the production of ε-rhodomyeinone in P1.
Similar content being viewed by others
References
Aszalos A. A., Bachur N. R., Hamilton B. K., Langlykke A. F., Roller P. P., Sheikh M. Y., Sutphin M. S., Thomas M. C., Wareheim D. A., Wright L. H.: Microbial reduction of the side-chain carbonyl of daunorubicin and N-acetyldaunorubicin.J. Antibiotics 30, 50 (1977).
Bachur N. R., Gee M.: Daunorubicinol metabolism by rat tissue preparations.J. Pharmacol. Exp. Ther. 177, 567 (1971).
Bachur N. R., Gee M.: Oxygen sensitive reductive cleavage of daunorubicin and adriamycin.Fed. Proc. 31, 835 (1972).
Bachur N., Gee M.: Microsomal reductive glycosidase.J. Pharmacol. Exp. Ther. 197, 681 (1976).
Blumauerová M., Matějů J., Stajner K., Vaněk Z.: Studies on the production of daunomycinone-derived glycosides and related metabolites inStreptomyces coeruleorubidus andStreptomyces peucetius.Folia Microbiol. 22, 275 (1977).
Blumauerová M., Pokorný V., Šťastná J., Hošťálek Z., Vaněk Z.: Improved yields of daunomycinone glycosides in developmental mutants ofStreptomyces coeruleorubidus.Folia Microbiol. 23, 249 (1978a).
Blumauerová M., Stajner K., Pokorný V., Hošťálek Z., Vaněk Z.: Mutants ofStreptomyces coeruleorubidus impaired in the biosynthesis of daunomycinone glycosides and related metabolite.Folia Microbiol. 23, 255 (1978b).
Blumauerová M., Královcová E., Hošťálek Z., Vaněk Z.: Intra- and interspecific cosynthetic activity of mutants ofStreptomyces coeruleorubidus andStreptomyces galilaeus impaired in the biosynthesis of anthracyclines.Folia Microbiol. 24, 128 (1979).
Bradler G., Eckardt K., Fügner R.: Zur Charakterisierung des Streptomyces-Stammes JA 3043 und Gewinnung von Galirubin.Z. Allg. Mikrobiol. 6, 361 (1966).
Huffman D. H., Bachur N. R.: Daunorubicin metabolism in acute myelocytic leukemia.Blood 39, 637 (1972).
Jizba J., Sedmera P., Vokoun J., Blumauerová M., Vaněk Z.: Naphthacenequinone derivatives from a mutant strainStreptomyces coeruleorubidus.Coll. Czech. Chem. Commun. 44, in press (1979).
Karnetová J., Matějů J., Sedmera P., Vokoun J., Vaněk Z.: Microbial transformation of daunomycinone byStreptomyces aureofaciens B-96.J. Antibiotics 29, 1199 (1976).
Kern D. L., Bunge R. H., French J. C., Dion H. W.: The identification of ε-rhodomycinone and 7-deoxy- daunorubicinol aglycone in daunorubicin beers.J. Antibiotics 30, 432 (1977).
Komiyama T., Matsuzawa Y., Oki T., Inui T., Takahashi Y., Naganawa H., Takeuchi T., Umezawa H.: Baumycins, new antitumor antibiotics related to daunomycin.J. Antibiotics 30, 619 (1977).
Královcová E., Blumauerová M., Vaněk Z.: Strain improvement inStreptomyces galilaeus, a producer of anthracycline antibiotics galirubins.Folia Microbiol. 22, 321 (1977a).
Královcová E., Tax J., Blumauerová M., Vaněk Z.: Bewertung der Produktion von Glycosiden des ε-Pyrromycinons (Galirubine) bei der Züchtung des StammesStreptomyces galilaeus.Z. Allg. Mikrobiol. 17, 47 (1977b).
Marshall V. P., Reisender E. A., Reinere L. M., Johnson J. H., Wiley P. F.: Reductive microbial conversion of anthracycline antibiotics.Biochemistry 15, 4139 (1976a).
Marshall V. P., Reisender E. A., Wiley P. F.: Bacterial metabolism of daunomycin.J. Antibiotics 29 966 (1976b).
Matéjů J., Vokoun J., Blumauerová M., Vaněk Z.: 7-Deoxy-13-dihydrodaunomycinone in cultures ofStreptomyces coeruleorubidus.Folia Microbiol. 23, 246 (1978).
Matéjů J., Blumauerová M., Vaněk Z.: Activity of enzymes transforming daunomycin inStreptomyces coeruleorubidus.Folia Microbiol. 24, 000 (1979).
McCarville M., Marshall V.: Partial purification and characterization of a bacterial enzyme catalyzing reductive cleavage of anthracycline glycosides.Biochem. Biophys. Res. Commun. 74, 331 (1977).
Ninet L., Florent J., Lunel J., Renault J., Abraham A., Lombardi B., Tissier R.: Duborimycin (20798 RP) — preparation by biochemical reduction of daunorubicin. 5th Internat. Ferment. Symp. Berlin. Abstracts of Papers (H. Dellweg, Ed.), p. 320 (1976).
Oki T., Komiyama T., Tone H., Inui T., Takeuchi T., Umezawa H.: Reductive cleavage of anthracycline glycosides by microsomal NADPH-cytochromec reductase.J. Antibiotics 30, 613 (1977).
Paranosenkova V. I., Karpov V. L.: Rubomycin biosynthesis. Glycosylation of exogenic rubomycinone and carminomycinone by rubomycin-producing organism. (In Russian)Antibiotiki 22, 37 (1977).
Takahashi Y., Naganawa H., Takeuchi T., Umezawa H., Komiyana T., Oki T., Inui T.: The structure of baumyeins A1, A2, B1, B2, C1 and C2.J. Antibiotics 30, 622 (1977).
Vaněk Z., Tax J., Komersová I., Eckardt K.: Glycosidation of ε-pyrromycinone using the strainStreptomyces galilaeus JA 3043.Folia Microbiol. 18, 524 (1973).
Vaněk Z., Tax J., Komersová I., Sedmera P., Vokoun J.: Anthracyclines.Folia Microbiol. 22, 139 (1977).
Wiley P. F., Marshall V. P.: Microbial conversion of anthracycline antibiotics.J. Antibiotics 28, 838 (1975).
Wiley P. F., Koert F. H., Elrod D. M., Reisender E. A., Marshall V. P.: Bacterial metabolism of anthracycline antibiotics. Steffimycinone and steffimycinol.J. Antibiotics 30, 649 (1977).
Author information
Authors and Affiliations
Additional information
ε-Rhodomycinone, 13-dihydrodaunomycinone and 7-deoxy-13-dihydrodaunomycinone were also found in other strains producing glycosides of daunomycinone (Kernet al. 1977; Komiyamaet al. 1977). Sugar components of glycosides isolated fromS. coeruleorubidus ME 130-A 4 were identified by Takahashiet al. (1977).
The author wish to thank Dr. W. Koch (Central Institute for Microbiology and Experimental Therapy, Jena, GDR) for the sample of ε-isorhodomycinone and Dr. P. Sedmera and Dr. J. Vokoun (Institute of Microbiology, Czechoslovak Academy of Sciences, Prague) for the measurement of NMR and mass spectra confirming the identity of the isolated anthracyclinones with the standards.
Rights and permissions
About this article
Cite this article
Blumauerová, M., Královcová, E., Matějů, J. et al. Biotransformations of anthracyclinones inStreptomyces coeruleorubidus andStreptomyces galilaeus . Folia Microbiol 24, 117–127 (1979). https://doi.org/10.1007/BF02927295
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02927295