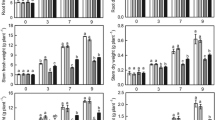
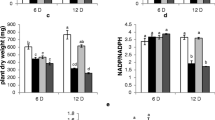
Abstract
The enzyme activities of the proline metabolic pathways were determined in control and satt-treated (140 M NaCl) calli derived from cotyledons of the domestic saltsensitive tomatoLycopersicon esculentum and the wild salt-tolerantL. pennellii. Glutamate, glutamine, asparagine, and aspartate levels increased in both genotypes under salt stress, while proline accumulation increased markedly only in the salt sensitive tomato. Activity of glutamine synthetase (GS) decreased in the salt-treated calli of the domestic species, whereas both NADH- and NADPH-glutamate synthase (GOGAT) activities increased; GS and NADPH-GOGAT decreased together in the salinized calli of the wild species. Decreasing ornithine levels were found due to NaCl in both tomato populations, while ornithine transaminase (OT) decreased in the wild type only. Increasing NADPH-Δ-pyrroline-5-carboxylate reductase (P5CR) and decreasing proline oxidase (Pro oxi) occurred in the salinized calli of the wild type. Conversely, Pro oxi and proline dehydrogenase (Pro dH) decreased highly in the salinized calli of the domestic population, while no significant changes in P5CR were found.
Similar content being viewed by others
Abbreviations
- DCPIP:
-
dichlorophenol 2,6 indophenol
- DTT:
-
dithiotritol
- FAD:
-
flavin adenine dinucleotide
- GOGAT:
-
glutamate synthase
- GS:
-
glutamine synthetase
- OT:
-
ornithine transaminase
- P5CR:
-
Δ-pyrroline-5-carboxylate reductase
- Pro dH:
-
proline dehydrogenase
- Pro oxi:
-
proline oxidase
- RGR:
-
relative growth rate
References
Bates, L.S., Waldren, R.P., Teare, I.D.: Rapid determination of free proline for water stress studies.— Plant Soil39: 205–207, 1973.
Bellinger, Y., Larher, F.: Proline accumulation in higher plants: a redox buffer?.—Plant Physiol. (Life Sci. Adv.)6: 23–27, 1987.
Boggess, S.F., Aspinall, D., Paleg, L.G.: Stress metabolism IX. The significance of end-products inhibition of proline biosynthesis and compartmentation in relation to stress induced proline accumulation.—Aust. J. Plant Physiol.3: 513–526, 1976a.
Boggess, S.F., Stewart, C.R., Aspinall, D., Paleg, L.G.: Effect of water stress on proline synthesis from radioactive precursors.—Plant Physiol.58: 398–401, 1976b.
Bourgeais-Chaillou, P., Guerrier, G.: Salt-responses inLycopersicon esculentum calli and whole plants.—J. Plant Physiol.140: 494–501, 1992.
Charest, C., Phan, C.T.: Cold acclimation of wheat: properties of enzymes involved in proline metabolism.—Physiol. Plant.80: 159–168, 1990.
Delaunay, A.J., Verna, D.P.S.: A soybean Δ-pyrroline-5-carboxylate reductase gene was isolated by functional complementation inEscherichia coli and is found to be osmoregulated.—Mol. gen. Genet.221: 299–305, 1990.
Golan-Goldhirsch, A., Hankamer, B., Lips, S.H.: Hydroxyproline and proline content of cell walls of sunflower, peanut and cotton grown under salt stress.—Plant. Sci.69: 27–32, 1990.
Golan-Goldhirsch, A., Hankamer, B., Lips, S.H.: Hydroxyproline and proline content of cell walls of sunflower, peanut and cotton grown under salt stress.—Plant Sci.69: 27–32, 1990.
Huang, A.H., Cavalieri, A.J.: Proline oxidase and water stress induced proline accumulation in spinach leaves.—Plant Physiol.63: 531–535, 1979.
Laliberté, G., Hellebust, J.A.: Pyrroline-5-carboxylate reductase inChorella autotrophica andC. saccharophila in relation to osmoregulation.—Plant Physiol.91: 917–923, 1989.
LaRosa, P.C., Rhodes, D., Rhodes, J.C., Bressan, R.A., Csonkas, L.N.: Elevated accumulation of proline in NaCl-adapted tobacco cells is not due to altered pyrroline-5-carboxylate reductase.—Plant Physiol.96: 245–250, 1991.
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J.: Protein measurement with the Folin phenol reagent.—J. biol. Chem.193: 265–275, 1951.
Munns, R., Termaat, A.: Whole plant response to salinity.—Aust. J. Plant Physiol.13: 143–160, 1986.
Nash, D., Paleg, L.G., Wiskich, J.T.: Effect of proline, betaine and some other solutes on the heat stability of mitochondrial enzymes.—Aust. J. Plant Physiol.9: 47–54, 1982.
Noguchi, M., Koiwai, A., Tanaki, E.: Studies on nitrogen metabolism in tobacco plant.—Agr. Biol. Chem.30: 452–456, 1966.
Rayapati, P.J., Stewart, C.R., Hack, E.: Pyrroline-5-carboxylate reductase is in pea leaf chloroplasts. —Plant Physiol.91: 581–586, 1989.
Rhodes, D., Handa, S., Bressan, R.A.: Metabolic changes associated with adaptation of plant cells to water stress.—Plant Physiol.82: 890–903, 1986.
Stewart, C.R., Boggess, S.F.: The effect of wilting on the conversion of arginine, ornithine and glutamate to proline in bean leaves.—Plant Sci. Lett.8: 147–153, 1977.
Stewart, C.R., Boggess, S.F., Aspinall, D., Paleg, L.G.: Inhibition of proline oxidase by water stress.—Plant Physiol.59: 930–932, 1977.
Steward, G.R., Larher, F.: Accumulation of amino acids and related compounds in relation to environmental stress.—In: Miflin, B.J. (ed.): Biochemistry of Plants. A Comprehensive Treatise. Vol. 5. Amino Acids and Derivatives. Pp. 609–632. Academic Press, New York 1980.
Tal, M., Katz, A.: Salt tolerance in the wild relatives of the cultivated tomato: the effect of proline on the growth of callus tissue ofLycopersicon esculentum andL. peruvianum under salt and water stress.—Z. Pflanzenphysiol.98: 283–288, 1980.
Tal, M., Katz, A., Heikin, H., Dehan, K.: Salt tolerance in the wild relatives of the cultivated tomato: proline accumulation inLycopersicon esculentum, L. peruvianum andL. pennellii treated with NaCl and PEG.—New Phytol.82: 349–355, 1979.
Treichel, S.: The influence of NaCl on pyrroline-5 carboxylate reductase in proline-accumulating cell suspension cultures ofMesembryanthemum nodiflorum and other halophytes.—Physiol. Plant.67: 173–181, 1986.
Venekamp, J.H.: Regulation of cytosol acidity in plants under conditions of drought—Physiol. Plant.76: 112–117, 1989.
Author information
Authors and Affiliations
Additional information
communicated by I. MACHÁČKOVÁ
Rights and permissions
About this article
Cite this article
Rus-Alvarez, A., Guerrier, G. Proline metabolic pathways in calli fromLycopersicon esculentum andL. pennellii under salt stress. Biol Plant 36, 277–284 (1994). https://doi.org/10.1007/BF02921101
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02921101