Abstract
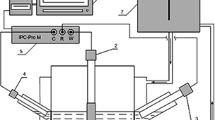
The rate of extraction of copper (II) from acid nitrate solutions with LIX 64N was studied in a single drop reaction cell in which experimental conditions were conrrolled so that the back reaction kinetics were unimportant. Under these conditions the extraction reaction was found to be first order with respect to the cupric ion concentration, half order with respect to the concentration of the α-hydroxy oxime component of LIX 64N, and half order with respect to the concentration of the hydroxy-benzophenone oxime component of LIX 64N. Detailed consideration of the experimental results and the hydrodynamics of a falling drop system suggests that a surface reaction mechanism is rate controlling and that the reaction rate is not limited by mass transfer. The apparent activation enthalpy of approximately 6.5 kcal/mole (27.2 kJ/mole) was found to be essentially independent of the variables considered.
Similar content being viewed by others
References
R. L. Atwood and J. D. Miller,Trans. SME AIME, 1973, vol. 254, p. 319.
Bagdad Copper Corp.,Liquid ion Exchange and Electrowinning Plant, Bagdad Copper Corp., Bagdad Arizona, 1971.
R. Scheffei: Private Communication, Miami, Arizona, June, 1973.
Large Chingola Copper Solvent Extraction Plant Seen as Major Advance, Eng. Mining J., July, 1973, vol. 174, p. 21.
W. Nitsch:Ber. Bungsenges., 1965, vol. 60, pp. 884–93.
F. H. Garner and M. Tayeban:An. Real Soc. Expan. Fis. Quim., 1960, vol. 56B, p. 479.
L. Crocker, R. O. Dannenberg, D. W. Bridges, and J. B. Rosenbaum:Recovery of Beryllium from Spor Mountain, Utah Ore by Solvent Extraction and Caustic Stripping, USBM RI 6173, 1962.
D. R. Spink and D. N. Okuhava:Comparative Equilibrium and Kinetics of KELEX 100/120 and LIX 63/65N/64N Systems for Extraction of Copper, p. 497, International Symposium on Hydrometallurgy, AIME, 1973.
R. Neelamegghan:Solvent Extraction Studies with Hydroximes and Alkylphosphoric Acid, Ph.D. Thesis, University of Utah, Salt Lake City, Utah, 1972.
R. R. Swanson:Compositions Containing Phenolic Oximes and Certain α-Hydroxy Aliphatic Oximes, U.S. Patent 3,592,775, July 13, 1971.
A. D. Ryon, F. L. Daley, and R. S. Lowrie:Design and Scaleup of Mixer-Settlers for the Dapex Solvent Extraction Process, Oak Ridge National Laboratory Report ORNL-2951,74 pp., 1960.
A. W. Ashbrook:Anal. Chim. Acta, 1972, vol. 58, p. 115.
F. H. Garner:Trans. Inst. Chem. Eng., London, 1950, vol. 28, p. 88.
R. C. Kintner:Advances in Chemical Engineering, Drew, ed., vol. 4, p. 51, Academic Press, 1963.
P. M. Heerfjes and L. H. De Nie:Recent Advances in Liquid-Liquid Extraction, Hanson, ed., p. 367, Pergamon Press, 1971.
J. T. Davies:Advances in Chemical Engineering, Drew, ed., vol. 4, p. 1. Academic Press, 1963.
G. V. Levich:Physicochemical Hydrodynamics, p. 404 Prentice-Hall Inc., Englewood Cliffs, N. J., 1962.
R. H. Magarvey and C. S. Maclatchy:A. I. Ch.E., 1968, vol. 14, no. 21, p. 263.
K. F. Reid:Properties and Reactions of Bonds in Organic Molecules, p. 469, Elsevier, N. Y., 1968.
D. S. Flett, D. N. Okuhara, and D. R. Spink:J. Inorg. Nucl. Chem., 1973, vol. 35, p. 2471.
J. D. Miller and R. L. Atwood:J. Inorg. Nucl. Chem., accepted for publication February 1975.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Atwood, R.L., Thatcher, D.N. & Miller, J.D. Kinetics of copper extraction from nitrate solutions by LIX 64N. Metall Trans B 6, 465–473 (1975). https://doi.org/10.1007/BF02913834
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02913834