Summary
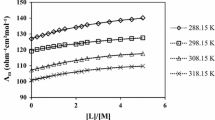
Rates of the solvolysis of [Co(CN)5Cl]3− ions have been determined in mixtures of water with the hydrophobic alcohol, t-butanol over a range of temperatures. No linear correlation of log k with the reciprocal of the dielectric constant is found, suggesting that changes in solvent structure are an important factor influencing these rates. This result is confirmed by the good correlation found for the extrema in the enthalpy and entropy of activation with the extrema in the physical properties of the mixtures influenced by changes in solvent structure. The application of a free energy cycle to the loss of the chloride ion from the Co3+ in the transition state shows that [Co(CN)5]2− in the transition state is stabilised in the mixtures relative to [Co(CN)5Cl]3− in the initial state.
Similar content being viewed by others
References
C. N. Elgy and C. F. Wells,J. Chem. Soc. Dalton Trans., 2405 (1980); 1617 (1982);J. Chem. Soc. Faraday Trans. 1,79, 2367 (1983).
I. M. Sidahmed and C. F. Wells,J. Chem. Soc. Dalton Trans., 1035 (1983); 1969 (1984); I. M. Sidahmed and A. M. Ismail,Transition Met. Chem.,11, 288 (1986).
K. H. Halawani and C. F. Wells,J. Chem. Res., (S) 58, (M) 0501 (1988).
D. A. Bush and C. F. Wells,J. Chem. Soc. Farad. Trans.,86, 941 (1990).
I. M. Sidahmed and A. M. Ismail.Transition Met. Chem.,12, 332, 449 (1987).
G. M. El-Subruiti, I. M. Sidahmed and C. F. Wells,Int. J. Chem. Kin.,22, 891 (1990).
K. H. Halawani and C. F. Wells,J. Chem. Soc. Farad. Trans. I,85, 2999 (1989);J. Solution Chem.,11, 1073 (1990).
K. Nakanishi,Bull. Chem. Soc. Jpn,33, 793 (1960).
M. J. Blandamer,Introduction to Chemical Ultrasonics, Academic Press, London, 1973, Chap. 11.
A. E. Dunstan,Zeit. Phys. Chem.,49, 590 (1904);51, 732 (1905).
G. Wada and S. Umeda,Bull. Chem. Soc. Jpn,35, 646 (1962).
W. E. Jones, L. R. Carey and T. W. Swaddle,Can. J. Chem.,50, 2739 (1972); D. A. Palmer and H. Kelm,Inorg. Chem.,16, 3139;Coord. Chem.,36, 39 (1981); G. Daffner, D. A. Palmer and H. Klem,Inorg. Chim. Acta,45, L275 (1980); G. A. Lawrance and S. Suvichittanont,Austral. J. Chem.,33, 273 (1980);Inorg. Chim. Acta,44, L61 (1980); M. J. Sisley and T. W. Swaddle,Inorg. Chem.,20, 2799 (1981); G. A. Lawrance,Inorg. Chem.,21, 3687 (1982); Y. Kitamara and R. van Eldik, Ber.Bunsen Ges. Phys. Chem.,88, 418 (1984); G. A. Lawrance, K. Schneider and R. van Eldik,Inorg. Chem.,23, 392 (1984); N. J. Curtis and G. A. Lawrance,Inorg. Chem.,25, 1033 (1986); P. A. Lay,Inorg. Chem.,26, 2144 (1987); N. J. Curtis, G. A. Lawrance and R. van Eldik,Inorg. Chem.,28, 329 (1989).
W. G. Jackson and A. M. Sargeson,Inorg. Chem.,17, 1344 (1978); W. G. Jackson and C. M. Begbie,Inorg. Chim. Acta,60, 115 (1982).
C. F. Wells,J. Chem. Soc. Farad. Trans. 1,73, 1851 (1977).
A. Haim and W. K. Wilmarth,Inorg. Chem.,1, 573, 583 (1962); A. Haim, R. J. Grassi and W. K. Wilmarth,Adv. Chem.,49, 31 (1966);Inorg. Chem.,6, 237 (1967); R. Barca, J. Ellis, M.-S. Tao and W. K. Wilmarth,Inorg. Chem.,6, 243 (1967); J. E. Byrd and W. K. Wilmarth,Inorg. Chim. Acta Rev.,5, 7 (1971).
A. Haim,Inorg. Chem.,21, 2887 (1981).
M. G. Burnett and M. W. Gilfillan,J. Chem. Soc. Dalton Trans., 1578 (1981); M. H. M. Abu-El-Wafa and M. G. Burnett,J. Chem. Soc. Chem. Comm. 833 (1983); M. H. M. Abou-El-Wafa, M. G. Burnett and J. F. McCullagh,J. Chem. Soc. Dalton Trans., 1059, 2311 (1987).
D. A. Palmer and H. Kelm,Zeit. anorg. allg. Chem.,450, 50 (1979).
K. H. M. Halawani and C. F. Wells,J. Chem. Soc. Farad. Trans. I,86, 1791 (1990).
K. J. Laidler and P. A. Landskroener,Trans. Farad. Soc.,52, 200 (1956).
T. L. Broadwater and R. L. Kay,J. Phys. Chem.,74, 3802 (1970).
A. C. Brown and D. J. G. Ives,J. Chem. Soc., 1608 (1962).
G. Åkerlöf,J. Amer. Chem. Soc.,54, 4125 (1932).
E. Grunwald and S. Winstein,J. Amer. Chem. Soc.,70, 846, (1948).
G. S. Groves and C. F. Wells,J. Chem. Soc. Farad. Trans. I 81, 2475 (1985).
J. Kenttämaa, E. Tommila and M. Martti,Annal. Acad. Scient. Fennicae, No. 93 (1959).
C. F. Wells,Trans. Faraday Soc.,66, 204 (1970);J. Chem. Soc., Farad. Trans. I,70, 694 (1974).
H. S. Frank and M. W. Evans,J. Chem. Phys.,13, 507 (1945); H. S. Frank and W.-Y. Wen,Discussion Farad. Soc.,24, 133 (1957); G. Nemethy and H. A. Sheraga,J. Chem. Phys.,36, 3382, 3401 (1962); N. Laiden and G. Nemethy,J. Phys. Chem.,74, 3501, (1970).
J. H. Andreae, P. D. Edmonds and J. F. McKellar,Acustica,15, 74 (1965).
G. S. Groves and C. F. Wells,J. Chem. Soc. Farad. Trans. I,78, 619 (1982); A. E. Eid and C. F. Wells,J. Chem. Soc. Farad. Trans. I,77, 1621 (1981);79, 253 (1983);81, 4101 (1985);82, 1643 (1986);Transition. Met. Chem.,10, 223 (1985);Int. J. Chem. Kin.,18, 215, (1986); G.S. Groves, A.F.M. Nazer and C.F. Wells,J. Chem. Soc. Farad. Trans. I,83, 1119 (1987).
C.F. Wells,J. Chem. Soc. Farad. Trans. I,72, 601, (1976);Australian J. Chem.,36, 1739 (1983).
J. Juillard and C. Tissier,Electrochim. Acta,27, 123 (1982).
T. A. Clune, D. Feakins and P. J. McCarthy,J. Electroanal. Chem.,84, 199 (1977).
Y. Pointud, J. Juillard, J.-P. Morel and L. Avedikan,Electrochim. Acta,19, 229 (1974).
C. F. Wells,Thermochim. Acta,132, 127, 141 (1988).
C. F. Wells,J Chem. Soc. Farad. Trans. I,80, 2445 (1984).
Author information
Authors and Affiliations
Additional information
TMC 2494
Rights and permissions
About this article
Cite this article
Halawani, K.H.M., Wells, C.F. Kinetics of the solvolysis of chloropentacyanocobaltate(III) ions in water +t-butanol mixtures. Transition Met. Chem. 17, 369–373 (1992). https://doi.org/10.1007/BF02910709
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02910709