Summary
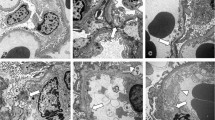
Characteristic pathological changes in the glomeruli in diabetic nephropathy include expansion of the mesangial matrix and thickening of the glomerular basement membrane (GBM). Using an acellular digestion technique combined with scanning electron microscopy, the three-dimensional ultrastructural changes in glomerular extracellular matrices were studied in rats with diabetic glomerulopathy. Diabetes was induced by the intravenous injection of streptozotocin and morphological analyses were performed 3, 6 and 11 months after the injection. Expansion of mesangial area and GBM thickening became evident with time. After treatment with the series of detergents, all cellular components were completely removed leaving the extracellular matrices intact. In normal controls, the mesangial matrix appeared as fenestrated septa with oval or round stomata between the glomerular capillaries. In diabetic glomerulopathy, expansion of mesangial matrix and narrowing of the mesangial fenestrae were observed. These changes in the mesangial matrices seem to play a vital role in the progression of glomerulosclerosis in rat diabetes. A subendothelial thin layer of the GBM was continuous with the mesangial matrix. One cause of GBM thickening in streptozotocin diabetes may be expansion of the mesangial matrix into the peripheral GBM.
Similar content being viewed by others
References
Beisswenger PJ, Spiro RJ (1970) Human glomerular basement membrane: chemical alteration in diabetes mellitus. Science 168:596–598
Bendayan M (1985) Alteration in the distribution of type IV collagen in glomerular basal laminae in diabetic rats as revealed by immunocytochemistry and morphometrical approach. Diabetologia 28:373–378
Bonsib SM (1985) Scanning electron microscopy of acellular glomeruli in nephrotic syndrome. Kidney Int 27:678–684
Bonsib SM (1988) Glomerular basement membrane necrosis and crescent organization. Kidney Int 33:966–974
Carlson EC, Kenney C (1980) Preparation and histoarchitecture of ultrastructurally pure basement membrane. Renal Physiol 3:280–287
Carlson EC, Hinds D (1983) A topographical (SEM) analysis of acellular glomerular mesangial matrix in situ. J Ultrastruct Res 82:96–110
Carlson EC, Surerus KK (1986) SEM studies of acellular glomerular basement membrane in human diabetic glomerulopathy. Anat Rec 216:349–358
Dachs S, Churg J, Mautner W, Grishman E (1964) Diabetic nephropathy. Am J Pathol 44:155–168
Ekblom P (1981) Formation of basement membranes in the embryonic kidney. An immunohistological study. J Cell Biol 91:1–10
Falk RJ, Scheiman JI, Mauer SM, Michael AF (1983) Polyantigenic expansion of basement membrane constituents in diabetic nephropathy. Diabetes [Suppl 2] 32:34–39
Fisher ER, Perez-Stable E, Amidi M, Sarver ME, Danowski TS (1967) Ultrastructural renal changes in juvenile diabetes. JAMA 202:291–295
Gundersen HJG, Jensen TB, Østerby R (1978) Distributions of membrane thickeness determined by lineal analysis. J Microsc 113:27–43
Heptinstall RH (1983) Diabetes mellitus and gout. In Pathology of the kidney, 3rd edition. Little Brown and Co Boston, pp 1397–1453
Huang TW (1979) Basal lamina heterogeniety in the glomerular capillary tufts of human kidneys. J Exp Med 149:1450–1459
Huang TW (1980) The nature of basal lamina alterations in human diabetic glomerulosclerosis. Am J Pathol 100:225–238
Kanwar YS, Rosenzweigh LJ, Linker A, Jakobowski ML (1983) Decreased de novo synthesis of glomerular proteoglycans in diabetes: Biochemical and autoradiographic evidence. Proc Natl Acad Sci USA 80:2272–2275
Kimmelstiel PG, Osawa G, Beres J (1966) Glomerular basement membrane in diabetes. Am J Clin Pathol 45:21–34
Makino H, Takaoka M, Komoda K, Tokuyama K, Ota Z, Ofuji T (1981) Leukocytes in the glomerular capillary lumen in Masugi nephritis. A scanning electron microscopic study of freezecracked specimens. Acta Med Okayama 35:373–376
Makino H, Nishimura S, Soda K, Takaoka M, Kaneshige T, Ota Z (1986) Mechanism of Hematuria I. Electron microscopic demonstration of the passage of a red blood cell through a glomerular capillary wall in rat Masugi nephritis. Virchows Arch [B] 50:199–208
Makino H, Nishimura S, Takaoka M, Ota Z (1988) Mechanism of hematuria. II. Scanning electron microscopic demonstration of the passage of blood cells through a glomerular capillary wall in rabbit Masugi nephritis. Nephron 50:142–150
Makino H, Ota Z (1989) Three-dimensional architecture of the mesangial matrix. Comparison of the intact and acellular glomerulus. Jpn J Nephrol 31:1039–1045
Mauer SM, Steffes MW, Ellis EN, Sutherland DER, Brown DM, Goetz FC (1984) Structural-functional relationships in diabetic nephropathy. J Clin Invest 74:1143–1155
Meezan E, Hjelle JT, Brendel K, Carlson EC (1975) A simple versatile nondisruptive method for isolation of morphologically pure basement membranes from several tissues. Life Sci 17:1721–1732
Nishimura S, Makino H, Ota Z (1989) Three-dimensional ultrastructural changes of acellular glomerular basement membrane in various types of human glomerulonephritis. Nephron 53:9–17
Scheinman JI, Tsai C (1984) Monoclonal antibody to type IV collagen with selective basement membrane localization. Lab Invest 50:101–112
Weidner N, Lorentz WB (1986a) Scanning electron microscopy of the acellular glomerular and tubular basement membrane in lupus nephritis. Am J Clin Pathol 85:135–145
Weindner N, Lorentz WB (1986b) Scanning electron microscopy of the acellular glomerular basement membranes in idiopathic membranous glomerulopathy. Lab Invest 54:84–92
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Makino, H., Yamasaki, Y., Hironaka, K. et al. Glomerular extracellular matrices in rat diabetic glomerulopathy by scanning electron microscopy. Virchows Archiv B Cell Pathol 62, 19–24 (1992). https://doi.org/10.1007/BF02899661
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02899661