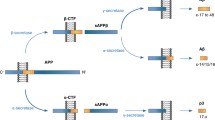
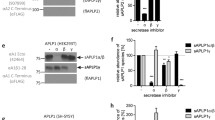
Abstract
Studies were undertaken on the processing of Alzheimer's disease-associated β-amyloid precursor protein in normal cultured human fibroblasts and a human neuroblastoma cell line. Major differences in processing between the secreted and intracellular forms of the precursor were found. The intracellular form appears to undergo amino-terminal processing yielding many smaller fragments, whereas the secreted from does not show any further proteolytic cleavage after its release from the cell surface. In pulse-chase experiments, antibodies to the A4 region immunoprecipitated bands of Mr=92,000−128,000, which represent the intact precursor; several smaller intracellular fragments of Mr=70,000–72,000, 55,000, 33,000 and 6,000 also immunoprecipitated with this antibody. The Mr=6,000 band cleared from the cell very quickly and is postulated to be the A4-carring remnant of the secreted protein. The data show that a fragment of Mr=33,000, which includes the A4 region, is one stable processed end-product of the intracellular precursor protein. It is possible that different posttranslational modifications are the signals responsible for the differences in processing between the secreted and intracellular amyloid precursor protein.
Similar content being viewed by others
References
Bancher, C., Grundke-Iqbal, I., Iqbal, K., Kim, K.S., Wisniewski, H.M. (1988). Immunoreactivity of neuronal lipofuscin with monoclonal antibodies to the amyloid beta protein. Alzheimer's Dis. Assoc. Disord. 2(3):262
Bittner, M., Kupfere, P., Morris, C.F. (1980). Electrophoretic transfer of protein and nucleic acids from slab gels to diazobenzyloxymethyl cellulose or nitrocellulose sheets.Anal. Biochem. 102:459–471
Bonner, W.M., and Laskey, R.A. (1974). A film detection method for tritium labelled proteins and nucleic acids in polyacrylamide gels. Eur. J. Biochem. 46:83–88
deWet, J.R., Fukushima, H., Dewji, N.N., Wilcox, E., O'Brien, J.S., Helinski, D.R. (1984). Chromogenic immunodetection of human serum albumin and alpha-l-fucosidase clones in a human hepatoma cDNA expression library. DNA 3:437–447
Dyrks, T., Weidemann, A., Multhaup, G., Salbaum, J.M., Lemaire, H.G., Kang, J., Muller-Hill, B., Masters, C.L., Beyreuther, K. (1988). Identification, transmembrane orientation and biogenesis of the amyloid A4 precursor of Alzheimer's disease. EMBO J. 7:949–957
Glenner, G.G. (1983). Banbury Report, 15: Biological Aspects of Alzheimer's Disease. R. Katzman (ed). Cold Spring Harbor Laboratory, Cold Spring Harbor, NY, pp. 137–144
Glenner, G.G., Wong, W.C. (1984). Alzheimer's disease: Initial report of the purification and characterisation of a novel cerebrovascular amyloid protein. Biochem. Biophys. Res. Commun. 120:885–890
Goldgaber, D., Lerman, M.I., McBride, O.W., Saffiotti, U., Gajdusek, D.C. (1987). Characterisation and chromosomal localisation of a cDNA encoding brain amyloid of Alzheimer's disease. Science 238:877–880
Kang, J., Lemaire, H.G., Unterbeck, A., Salbaum, J.M., Masters, C.L., Grzeschik, K.H., Multhaup, G., Beyreuther, K., Muller-Hill, B. (1987). The precursor of Alzheimer's disease amyloid A4 protein resembles a cell-surface receptor. Nature 325:733–736
Kitaguchi, N., Takahashi, Y., Shiojiri, S., Ito, H. (1988). Novel precursor of Alzheimer's disease amyloid protein shows protease inhibitory activity. Nature 331:530–532
Laemmli, U.K. (1970). Cleavage of structural proteins during the assembly of the head bacteriophage T4. Nature 227:680–685
Landon, M., Smith, M.A., Clarke, K., Kidd, M. (1988). Immunoaffinity chromatographic purification of amyloid related proteins from Alzheimer's disease brain. Alzheimer's Dis. Assoc. Disord. 2(3):261
Lerner, R.A., Green, H., Alexander, H., Liu, F.-T., Sutcliffe, J.G., Shinnick, T.M. (1981). Chemically synthesized peptides predicted from the nucleotide sequence of the hepatitis B virus genome elicit antibodies reactive with the native envelope protein of Dane particles. Proc. Natl. Acad. Sci. U.S.A. 78:3403–3407
Lynch, D.R., Snyder, S.H. (1986). Neuropeptides: Multiple molecular forms, metabolic pathways and receptors. Annu. Rev. Biochem. 55:773–779
Masters, C.L., Multhaup, G., Simms, G., Pottgiesser, J., Martins, R.N., Beyreuther, K. (1985a). Neuronal origin of a cerebral amyloid: Neurofibrillary tangles of Alzheimer's disease contain the same protein as the amyloid of plaque cores and blood vessels. EMBO J 4:2757–2763
Masters, C.L., Simms, G., Weinman, N.A., Multhaup, G., McDonald, B.L. (1985b). Amyloid plaque core protein in Alzheimer's disease and Down's syndrome. Proc. Natl. Acad. Sci. U.S.A. 82:4245–4249
Mita, S., Sadlock, J., Herbert, J., Schon, E.A. (1988). A cDNA specifying the human amyloid B precursor protein (ABPP) encodes a 95-kDa polypeptide. Nucleic Acids Res. 16:9351
Nestor, J.J., Tahilramani, R., Ho, T.L., McRae, G.I., and Vickery B.H. (1988). Potent, long-acting luteinizing hormone-releasing antagonists containing new synthetic amino acids:N,N′-dialkyl-d-homoarginines. Med. Chem. 31:65–72
O'Brien, J.S., Kretz, K.A., Dewji, N., Wenger, D.A., Esch, F., Fluharty, A. (1988). Coding of two sphingolipid activator proteins (SAP-1 and SAP-2) by the same genetic locus. Science 241:1017–1132
Ponte, P., Gonzalez-DeWhitt, P., Schilling, J., Miller, J., Hsu, D., Greenberg, B., Davis, K., Wallace, W., Lieberberg, I., Fuller, F., Cordell, B. (1988). A new A4 amyloid mRNA contains a domain homologous to serine proteinase inhibitors. Nature 331:525–527
Robakis, N.K., Anderson, J.P., Refolo, L.M., Haroutunian, V., Bierer, L., Perl, D., Salton, S. (1989). Differential expression of the Alzheimer's amyloid precursor protein in brain and cell-cultures. Alzheimer's Dis. Assoc. Disord. 3(Suppl 1):39
Selkoe, D.J., Podlisney, M.B., Joachim, C.L., Vickers, E.A., Lee, G., Fritz, L.C., Oltersdorf, T. (1988). B-amyloid precursor protein of Alzheimer's disease occurs as a 110–135 kilodalton membrane-associated protein in neural and non-neural tissues. Proc. Natl. Acad. Sci. U.S.A. 85:7341–7345
Shelton, E.R., Cohn, R., Fish, L., Obernolte, R., Tahiramani, R., Nestor, J., Chan, H.W. (1989). J. Neurochem. submitted
Tanzi, R.E., Gusella, J.F., Watkins, P.C., Bruns, G.P., St. George-Hyslop, P., Van Keuren, M.L., Patterson, D., Pajan, S., Kurnit, D.M., Neve, R.L. (1987). Amyloid beta protein gene: cDNA, mRNA distribution and genetic linkage near the Alzheimer's locus. Science 235:880–884
Tanzi, R.E., McClatchey, A.I., Lamberti, E.D., Villa-Komaroff, L., Gusella, J.F., Neve, R.L. (1988). Protease inhibitor domain encoded by an amyloid protein precursor mRNA associated with Alzheimer's disease. Nature 331:528–530
Terry, R.D. (1981). Dementia of the Alzheimer's type. Ann. Neurol. 10:184–192
Wiedemann, A., Konig, G., Bunke, D., Fischer, P., Salbaum, J.M., Masters, C.L., Beyreuther, K. (1989). Identification, biogenesis and localisation of precursors of Alzheimer's disease A4 amyloid protein. Cell 57:115–126
Author information
Authors and Affiliations
Additional information
This work was supported by grants from the National Institutes of Health (NS27580-01) and American Federation of Aging Research to N. Dewji, and grants PHS AR16322 and AG00402 to J.E. Seegmiller. M. Adler is a recipient of a Physician-Scientist Fellowship grant AG00353 to J.E. Seegmiller. The work was also supported by generous donations from the Patrons of Research of the Institute for Research on Aging, Mrs. Roberta Seegmiller, the Paul Glenn Foundation, and the Greenwall Foundation.
Rights and permissions
About this article
Cite this article
Dewji, N.N., Shelton, E.R., Adler, M.J. et al. Processing of Alzheimer's disease-associated β-amyloid precursor protein. J Mol Neurosci 2, 19–27 (1990). https://doi.org/10.1007/BF02896922
Issue Date:
DOI: https://doi.org/10.1007/BF02896922