Abstract
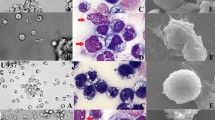
To investigate the antitumor activity of IL-12, the induction of differentiation of IL-12 was observed using erythroleukemia cells (FBL-3) as model. After incubation with 200 U/mL IL-12 for 48 h, DNA synthesis of FBL-3 cells in S phase decreased significantly; the expression of CD14 which is the specific marker of monocyte increased, the rate of NBT+ cells was apparently higher than that of the untreated FBL-3 cells. After treating FBL-3 cells with IL-12 for 72 h, the expression of 33D1 and NLDC145 which are the specific markers of dendritic cells increased markedly, the surface molecules such as MHC-11, B7-1, B7-2, and VCAM-1 were up-regulated; morphological observation showed two kinds of cells: some cells had a ruffled surface and plentiful lysosome; the others had many dendritic projections on the surface, and contained numerous mitochondria. Functionally, the IL-12-treated FBL-3 cells could apparently stimulate the proliferation of allogeneic and autologous T lymphocytes, and improve the specific cytotoxic activity of CTL on FBL-3 cells. These results indicated that erythroleukemia cells were induced by IL12 to differentiate into the monocytes and dendritic cells, then exhibited the antigen-presenting function. The data outline a new mechanism for IL-12 to treat leukemia.
Similar content being viewed by others
References
Scott, P., IL-12 initiation cytokine for cell-mediated immunity,Science, 1993, 260: 496.
Trinchieri, G., Interleukin-12: a cytokine produced by antigen-presenting cells with immunoregulatory functions in the generation of T-helper cells type 1 and cytotoxic lymphocytes,Blood, 1994, 84:4008.
Collins, S.J., Ruscetti, F.W., Gallagher, R.E. et al., Normal functional characteristics of cultured human promyelocytic leukemia cells (HL-60) after induction of differentiation by dimethysulfoxide,J. Exp. Med., 1979, 149:969.
Brunda, M.J., Luistro, L., Warner, R. R. et al., Antitumor and antimetastatic activity of interleukin 12 against murine tumors,J. Exp. Med., 1993, 178:1223.
Thierfelder, W.E., Deursen, J.M., Yamamoto, K. et al., Requirement for Stat 4 in interleukin-12-mediated responses of natural killer and T cells,Nature, 1996, 382: 171.
Pickl, W.F., Majdic, O., Kohl, P. et al., Molecular and functional characteristics of dendritic cells generated from highly purified CD14+ peripheral blood monocytes,J. Immunol., 1996, 157:3850.
Akagawa, K. S., Takasuka, N., Nozaki, Y. et al., Generation of CD1+ RelB+ dendritic cells and tartrateresistant acid phosphatase-positive osteoclast-like multinucleated giant cells from human monocytes,Blood, 1996, 88:4029.
Damle, N.K., Klussman, K., Linsley, P.S. et al., Differential costirnulatory effects of adhesion molecules B7, ICAM-1, LFA-3 and VCAM-1 on resting and antigen primed CD4+ T lymphocytes,J. Immunol., 1992, 148:1985.
Damle, N.K., Klussman, K., Leytze, G. et al., Costimulation of T lymphocytes with integrin ligands intercellular adhesion molecule-1 or vascular cell adhesion molecule-1 induces functional expression of CTLA-4, a second receptor for B7,J. Immunol., 1994, 152:2686.
Damle, N. K., Aruffo, A., Vascular cell adhesion molecular-1 induces T cell antigen receptor dependent activation of CD4+ T lymphocytes,Proc. Natl. Acad. Sci. USA, 1991, 88:6403.
Author information
Authors and Affiliations
Additional information
Project supported by the National High Biotechnology Foundation of China.
Rights and permissions
About this article
Cite this article
Zhao, Y., Cao, X. Immune activation of erythroleukemia cells induced by interleukin 12. Sci. China Ser. C.-Life Sci. 41, 323–329 (1998). https://doi.org/10.1007/BF02895109
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02895109