Abstract
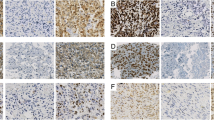
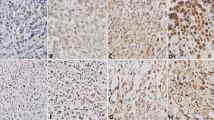
The p53 gene located at chromosome 17pl3 is found to be altered (allelic loss or other mutation) in multiple human cancers, including osteosarcomas. The mutated gene produces a protein with a prolonged half-life thus rendering it detectable by conventional immunohistochemistry. We examined the correlation between p53 expression and clinical prognosis as well as response to therapy. Twentyone patients with previously untreated and histologically verified highly malignant osteosarcoma were used for this study. Biopsy material taken both prior to the start of COSS 91 protocol and at the time of surgery (ten weeks later) was examined for alterations in p53 protein expression and drug resistance. Two patients who had strong (+++) p53 protein expression and three others who became positive during the chemotherapy had significantly worse prognosis (all of them died within one year) than those who showed no p53 expression both at biopsy and after chemotherapy (all 11 patients are alive, average follow-up time: 3.5 years). All patients who showed any kind of positive p53 protein expression on initial biopsy were non-respon-ders to chemotherapy. In contrast, 69% (9 out of 13) of those who exhibited no p53 expression on initial biopsy were responders or intermediate responders to chemotherapy. We concluded that p53 expression may be a useful prognostic factor in osteosarcomas. The direct correlation between p53 positive expression and resistance to therapy can help in identifying patients who are in need of a more vigorous or different chemotherapeutical protocol.
Similar content being viewed by others
References
Brunat-MentignyM and KohlerR: L’osteosarcome. Rev Prat 43: 2197–203, 1993.
Cattoretti G, Becker GM, KeyG, et al: Monoclonal antibodies against recombinant parts of the Ki-67 antigen (MIBI and MIB3) detect proliferating cells in microwave-processed formalin-fixed paraffin sections. Journal of Pathology 168: 357–363, 1992.
Chang F. Syrjanen S. Tervahauta A, and SyrjanenK: Tumourigenesis associated with the p53 tumor suppressor gene. Br J Cancer 68: 653–61, 1993.
Cordon-Cardo C, Lettres E, DrohnjakM, et al: Molecular abnormalities of mdm2 and p53 genes in adult soft tissue sarcomas. Cancer Res 54: 794–9, 1994.
Cotran RS, et al: Neoplasia. In Cotran RS, Kumar V, Robbins SL, (eds.): Robbins Pathologic Basis of Disease. Philadelphia, WB. Saunders Company, 1989, pp. 239–305.
Enneking WF, Spanier SS, Goodman MA: A system for the surgical staging of musculoskeletal sarcoma. Clin Orthop 153: 106, 1980.
Fanner G, Bargonetti J, ZlutH, et al: Wild-type p53 activates transcription in vitro. Nature 358: 83–86, 1992.
Friedman JM and McGillivrayB: Genetic Paradigms in Human Disease. In Friedman JM, et al. (eds.): Genetics. Baltimore, Williams & Wilkins, 1992, pp. 130–4.
Grundmann E, Veda Y Schneider-StockR, and RoessnerA: New aspects of cell biology in osteosarcoma. Pathol Res Pract 191:563–70, 1995.
Haines DS, Landers JE, Engle LJ, and George DL: Physical and functional interaction between wild-type p53 and mdm2 proteins. Mol Cell Biol 14:1171–8, 1994.
Hall PA and Lane DP: P53 in tumour pathology: can we trust immunohistochemistry? -Revisited!. Journal of Pathology 172:1–4, 1994.
Hanna E, Bodrogi I, InstitorisE, and BakM: A p53 expresszió és a klinikai rezisztencia összefüggésének vizsgálata hererákokban. Orvosi Hetilap 137: 59–64, 1996. (In Hungarian only)
HungJ and AndersonR: p53 functions, mutations and sarcomas. Acta Orthop Scand 273: 68–73, 1997
Isfort RJ, Cody DB, LovellG, and Doersen CJ: Analysis of oncogenes, tumor suppressor genes, autocrine growth-factor production, and differentiation state of human osteosarcoma cell lines. Mol Carcinog 14: 170–8, 1995.
Knudson AG: Antioncogenes and human cancer. Proc Natl Acad Sci USA 90: 10914–21, 1993.
KopperL and SehestyenA: P53 (egy molekula, mely „végigsöpört” a daganat kutatáson). Magyar Onkologia 39: 159- 164, 1995. (In Hungarian only)
Lee JM, Ahrahamson JLA, KandelR, et al: Susceptibility to radiation-carcinogenesis and accumulation of chromosomal breakage in p53 deficient mice. Oncogene 9: 3731–3736, 1994.
Levine AJ, Momand J, and Finlay CA: The p53 tumor suppressor gene. Nature 351: 453–456, 1991.
Lipponen PK: Over-expression of p53 nuclear oncoprotein in transitional- cell bladder cancer and its prognostic value. Int J Cancer 53: 465–470, 1993.
Malawer M, Link M, and DonaldsonS: Sarcomas of Bone. In Devita V, Hellman S, RosenbergS (eds.): Cancer Principles and Practice of Oncology. Philadelphia, J.B. Lippincott Co., 1993, pp. 1509–1552.
Nishikawa T, Yamamoto T, MizunoK, et al: Expression of the p53 protein in human osteosarcoma. Nippon Seikeigeka Gakkai Zasshi 68:400–406, 1994.
Salzer-Kuntschik M, Delling G, Beron G, SigmundR: Morphological grades of regression in osteosarcoma after poly chemotherapy study COSS 80. J Cancer Res Clin Oncol (Suppl) 106:21, 1983.
Serra M, Scotlandi K, Manara MC, et al: Establishment and characterization of multi-drug resistant human osteosarcoma cell lines. Anticancer Res 13: 323–9, 1993.
Veda Y, Dockhorn-Dworniczak B, BlasiusS, et al: Analysis of mutant P53 protein in osteosarcomas and other malignant and benign lesions of bone. J Cancer Res Clin Oncol 119:172–8, 1993.
WinklerK and BielachS: Chemotherapy of osteosarcoma. Sem Orthop 3: 48–58, 1988.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PÁpai, Z., N. FÉja, C., N. Hanna, E. et al. P53 overexpression as an indicator of overall survival and response to treatment in osteosarcomas. Pathol. Oncol. Res. 3, 15–19 (1997). https://doi.org/10.1007/BF02893346
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02893346