Abstract
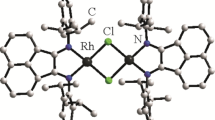
Two cis-dicahnyl rhodium complexes containing uncoordinated donors have been synthesized, and their IR and XPS data and the influence of their intramolecular substitution re-action on their stabilities have been reported. The intramolecular substitution reaction is re-versible. Under the condition of heating and without the protection of CO, the uncoordinated donors substitute the terminal carbonyls on the rhodium to form tetradentated complex. So the rhodium complex will not decompose for the drop of the terminal carbonyls and its stability is enhanced. In atmosphere of CO, it recovers to bidentate complex.
Similar content being viewed by others
References
Cramer, R., Olefin coordination compounds of rhodium (V)—The reactive stability and rates of exchange of olef in complexes of Rhodium (I),J. Am. Chem. Soc., 1976, 89: 4621.
Yuan, G. Q., Pan, P. L., Liu, Z. Y.et al., Prepation and catalytic activity of cis-[dicarbonyl(N-2(2-pyridyl) ethyllarylamine) rhodium] tetraphenyl borate,Journal of Molecular catalysis (in Chinese), 1995, 9(4): 278.
Wang, D. H., Liu, S. H., Yuan, G. Q., XPS study of square planar cis-dicarbonyl (bidentate K-donor ligand) rhodium (I) tetraphenyl borate,Acta Chimica Sinica (in Chinese), 1984, 42: 747.
Yin, G. C., Xi, Z. W., Chao, G. Y., Nickel complex with NO donors for catnlytic epoxidation of phenethylene,J. of Cat. (in Chinese), 1996, 17: 79.
Author information
Authors and Affiliations
About this article
Cite this article
Jiang, H., Pan, P. & Yuan, G. Influence of intramolecular substitution reaction of square planar carbonyl rhodium (I) cationic complex on its stability. Chin.Sci.Bull. 44, 1195–1198 (1999). https://doi.org/10.1007/BF02885964
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02885964