Abstract
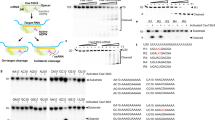
To study the expression activity of various vectors containing anti-caspase-3 ribozyme cassettesin vivo, and to further study the role of caspas-3 in the apoptotic pathway, we constructed anti-caspase-3 hammerhead ribozyme embedded into the human snRNA U6, and detected the activity of the ribozymein vitro andin vivo. Meanwhile we compared it with the self-cleaving hammerhead ribozymes that we previously studied, and with the general ribozyme, cloned into RNA polymerase II expression systems. The results showed that the three ribozymes, p1.5RZ107, pRZ107 and pU6RZ107 had the correct structure, and that they could cleave caspase-3 mRNA exactly to produce two fragments: 143nt/553nt. p1.5RZ107 has the highest cleavage efficiencyin vitro, almost 80%. However, the U6 chimeric ribozyme, pU6RZ107, has the highest cleavage activityin vivo, almost to 65%, though it has lower cleavage activityin vitro. The cleavage results demonstrated that the pU6RZ107, the U6 chimeric ribozyme, could more efficiently express and downregulate the level of caspase-3in vivo, and the ribozyme could provide an alternative approach to the research into the mechanism of apoptosis and human gene therapy also.
Similar content being viewed by others
References
Andrew, F., Gerard, E., A license to kill, Cell, 1996, 85:781–784.
Thornberry, N. A., Lazebnik, Y., Caspases: Enemies within, Science, 1998, 281: 1312–1316.
Kijima, H., Ishida, H., Ohkawa, T. et al., Therapeutic application of ribozymes, Pharmacol. Ther., 1995, 68: 247–264.
Phylactou, L. A., Kilpatrick, M. W., Wood, M. J., Ribozymes as therapeutic tools for genetic disease, Hum. Mol. Genet., 1998, 7(10): 1649–1653.
Bettrand, E., Pictet, R., Grange, T., Can heamerhead ribozymes be efficient tools inactivate gene function? Nucleic Acids Res., 1994, 22: 293–300.
Lieber, A., Strauss, M., Selection of efficient cleavage sites in target RNAs by using a ribozyme expression library, Mol. CellBiol., 1995, 15: 540–551.
Xu, R. H., Zhou, X. Q., Xie, Q. et al., Preparation and identification of hammerhead ribozymein vitro against rat cas-pase-3 mRNA fragment, Chin. J. Hepatol., 2000, 8: 361–363.
Liu, J., Jin, Y. X., Wang, D. B., A novel vector for abundant expression of antisense RNA, triplex-forming RNA and ribozymein vivo, High Technology Letters, 2000, 6: 84–88.
Sambrook, J., Fritsch, E. F., Maniatis, T., Molecular Cloning: A Laboratory Manual, 2nd ed., New York: Cold Spring Harbor Laboratory Press, 1989.
Porter, A. G., Jänicke, R. U., Emerging roles of caspase-3 in apoptosis, Cell Death Differ, 1999, 6: 99–104.
Cryns, V., Yuan, J., Proteases to die for, Genes Dev., 1998, 12: 1551–1570.
Narendra, K. V., Anikumar, R. K., Fritz, E., Recent developments in the hammerhead ribozyme field, Nucleic Acids Research, 1998, 26: 5237–5242.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, R., Liu, J., Xu, F. et al. Activity identification of chimeric anti-caspase-3 mRNA hammerhead ribozymein vitro andin vivo . Sci. China Ser. C.-Life Sci. 44, 618–627 (2001). https://doi.org/10.1007/BF02879356
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02879356