Abstract
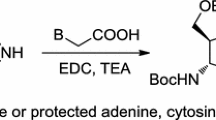
The physiologically active groups such as purine and pyrimidine bases are introduced to the asymmetric synthesis. The optically pure compounds bearing purine and pyrimidine bases (5a—5e) were prepared via the asymmetric Michael addition reaction of purines and pyrimidines as Michael donators with the chiral source 5-(R)-[(lR, 2S, 5R)-menthyloxy]-2(5H)-furanone (3a), which was prepared from the natural chiral auxiliary (-)-menthol. The synthetic method was studied in detail and the new compounds were identified on the basis of their analytical data and spectroscopic data, such as [α] 20D , IR, UV,1H NMR,13C NMR and MS. The absolute configuration of5a was established by X-ray crystallography. The results provided an efficient synthetic route to chiral purines and pyrimidine analogues, and offered chiral sources for further research on the physiologically active compounds of chiral nucleotides.
Similar content being viewed by others
References
United States Pharmacopoeia, Complete Drug Reference ed., New York: Consumer Reports Books, 1995, 175.
Keyano, S., Suzutani, T., Yoshida, I. et al., Analysis of phosphorylation pathways of perpesvirus nucleosides by varicellazoster virus-specific enzymes,Antimicrob Agents Chemother., 1996, 40: 920.
Liu Yugang, Fei Changpei, Study on the alkylation reaction of guanine and N-acetylguanine and the synthesis of 1’-C alkylated carba-DHPG and N-acetylguanine analogues.Chinese Journal of Chemistry, 1994, 12: 85.
Jiang Yunzhen, Tang Xiaolin, The synthesis of ganoder purine,Chinese Chemical Letters, 1995, 6: 661.
Van Oeveren, A., Jansen, J. F. G. A., Feringa, B. L., Enantioselective synthesis of natural dibenzybutyro-lactone lignans (-) -enteroctone, (-) -hinokinin, (-) -pluviatolide, (-) -enterodiol lignan (-) -eudesmi via tandem conjugate addition to γ-Alkoxybutenolides,J. Org. Chem., 1994, 59: 5999.
Pelter, A., Ward, R. S., Jones, D. M. et al., Asymmetric syntheses of lignans of the dibenzylbutyrolactone dibenzybutanediol aryltetralin and dibenzocyclooctadiene series,Tetrahedron: Asymmetry, 1992, 3: 239.
Feringa, B. L., Lange, B. de., Jong, C. de., Synthesis of enantiomerically pure (l-(Menthyloxy)-butenolides and (R) - and (S)-2-methyl-1, 4 butanediol,J. Org. Chem., 1989, 54: 2471.
Feringa, B. L., De. Jons, J. C., New strategies in asymmetric synthesis based on alkoxybutenolides,Bull. Soc. Chim. Belg., 1992, 101(7): 627.
Chen, Q. H., Geng, Z., Huang, B., Synthesis of a new chiral source 5-(l-menthyloxy)-2(5H)-furanone and the study of asymmetric Michael addition reaction,Youji Huaxue (in Chinese), 1991, 11: 494.
Chen, Q. H., Zou, C., Industrial synthesis of optically active compounds,Youji Yuaxue (in Chinese), 1994, 14: 1.
Chen, Q. H., Huang, B., Synthesis of borneol auxiliary chiral source and its stereospecific reaction,Chinese Science Bulletin (in Chinese), 1994, 39: 2154.
Chen, Q. H., Geng, Z., A new chiral source optically pure 5-(l-menthyloxy-3, 4-dichloro-2(5H)-furanone, its synthesis and structure,Acta Chimica Sinica (in Chinese), 1993, 51: 622.
Chen Qinghua, Gen Zhe., Haung Bin, Synthesis of enantiomerically pure 5-(l-menthyloxy)-3, 4-dibromo-2(5H)-furanone and its tandem asymmetric Michael addition-elimination reaction,Tetrahedron: Asymmetry, 1995, 6: 401.
Golinick, K., Griesheck, A., Singlet oxygen photooxygenation of furans,Tetrahedron, 1985, 41: 2057.
Lister, J. H., Fenn, M. D., “The Purities” (Supplement 1),Aninterscience Publication, New York: John Wiley, 1996, 257–261.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (Grant No. 29672004).
Rights and permissions
About this article
Cite this article
Wang, Y., Chen, Q. Synthesis of novel chiral compounds of purine and pyrimidine bases. Sc. China Ser. B-Chem. 42, 121–130 (1999). https://doi.org/10.1007/BF02875507
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02875507