Abstract
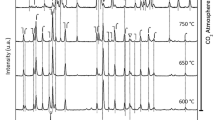
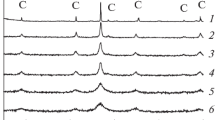
Two mixed oxide systems La2-xSrxCuO4± λ (0.0⩽x⩽1. 0) and La2-xThxCuO4± λ (O. O⩽x⩽ 0.4) with K2NiF4 structure were prepared by varyingx values. Their crystal structures were studied by means of XRD and IR spectra. The average valence of Cu ion at B site, nonstoichiometric oxygen (λ) and the chemical composition in the bulk and on the surface of the catalysts were measured by means of chemical analysis and XPS. The catalytic behavior in reaction CO+NO was investigated under the regular change of average valence of Cu ion at B site and nonstoichiometric oxygen (λ). Meanwhile, the adsorption and activation of the small molecules NO and the mixture of NO+CO over the mixed oxide catalysts were studied by means of MS-TPD. The catalytic mechanism of reaction NO+CO over these oxide catalysts were proposed; and it has been found that, at lower temperatures the activation of NO is the rate determining step and the catalytic activity is related to the lower valent metallic ion and its concentration, while at higher temperatures the adsorption of NO is the rate determining step and the catalytic activity is related to the oxygen vacancy and its concentration.
Similar content being viewed by others
References
Armor, J. N., Enviromental catalysis,Applied Catal., B, 1992, 1: 221.
Halase, I., Brenner, A., Shelef, M.et at., Decomposition of nitric oxide and its reduction by CO over superconducting and related cuprate catalysts,Catal. Lett., 1991, 11: 327.
Mizuno, N., Yamato, M.. Tanaka, M.et al., Reactions of CO and NO over La2- xA’xCu1-yB’yO4, a K2NiF4-type mixed oxide.Chem. of Materal., 1989, l(2): 232.
London, J. W., Bell, A. T., A simultaneous infrared and kinetic study of the reduction of nitric oxide by carbon monoxide over copper oxide,J. Cutal., 1973. 31: 96.
Cheng Tiexin, Yang Xiangguang, Wu Yue, Synthesis characterization and catalytic behaviour of La2-xSrxCoO4-λ(x = 0-2) in complex oxidation,Science in China, Ser. B, 1995, 38(9): 1025.
Zhao Zhen, Yang Xiangguang, Wu Yue, Comparative study of nickel-based perovskite-like mixed catalysts for direct decomposition of NO,Applied Catalysis, B:Environmental, 1996, 8: 281.
Zhao Zhen, Yang Xiangguang, Wu Yue, LaSrNiO4- λ with K2NiF4 structure: A highly active catalyst for direct decomposition of nitrogen monoxide,Chemical Research in Chinese University, 1996, 12(1): 81.
Zhao Zhen, Yang Xiangguang, Wu Yue, Direct decomposition of NO over the mixed oxide catalysts Nd2-xSrxNiO4 ± λ(0.0⩽ x⩽1.0),Chinese Science Bulletin, 1996. 41(11): 904.
Harris, D. C., Hewton, T. A., Determination of Cu3±/Cu2 + ratio in the superconductor YBa2Cu3O8-x,J. Solid State Chenl., 1987, 67: 182.
Wu, Y., Yu, T., Dou, B. S.et a1., A comparative study on perovskite-type mixed oxide catalysts A’xA1-xBO3- λ (A’= Ca, St, A=La, B=Mn, Fe, Co) for NH3 oxidation.J. Catal., 1989, 120: 88.
Tokura, Y., Takagi, H., Uchida, S., A superconducting copper oxide compound with electrons as the charge carriers,Nature, 1989, 337: 345.
Ogita, N., Udagawa, M., Kojima, K.et a1., Infrared and Raman study of (La1-xSrx)2NiO4 and La2(Ni1-xCux)xO4,J. Physical Society of Japan, 1989, 57(11): 3982.
Potts, W. J. Jr.,Chrnlical In frared Spectruscupy, Vol. 1, New York, London, Sydney: John Wiley, 1963, 135.
Arai, H., Adsorption of NOx on the surface of solid catalyst,Hyomen, 1976, 14: 427.
Hter Maat, J. H., Van Dijk, M. P., Rolelots, G., Electrical and catalytic properties of some oxides with the fluorite or pyrochlore structure, Part 2: Catalytic oxidation of carbon monoxide,Mat. Res. Bull., 1984, 14: 133.
Author information
Authors and Affiliations
Additional information
Project supported by the National Natural Science Foundation of China.
Rights and permissions
About this article
Cite this article
Zhao, Z., Yang, X. & Wu, Y. Characterization and catalytic behavior of La2 - x (Sr, Th)xCuO4 ± λ in reaction NO+CO. Sc. China Ser. B-Chem. 40, 464–474 (1997). https://doi.org/10.1007/BF02875414
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02875414