Abstract
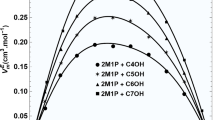
New isentropic compressibility data are reported for four ternary mixtures, which contained methylethylketone andn-octane as common components. 1-propanol, 1-butanol, 1-pentanol and 1-hexanol, which form a homologous series, were used as non-common components. The deviation in isentropic compressibility has been computed from the experimental results and compressibility of ideal mixtures. The deviation is positive over the whole range of volume fraction in the fourt mixtures. The positive deviation suggests that the structure breaking effect of the components outweighs the structure making effect.
Similar content being viewed by others
References
Naidu G R and Naidu P R 1981Acta Chim. 108 85
Naidu G R and Naidu P R 1982J. Chem. Engg. Data 27 57
Rao M V P 1974A study in thermodynamic and physical properties of binary liquid mixtures excess volumes isentropic compressibilities and viscosities Ph. D. Thesis, Sri Venkateswara University, Tirupati
Rao M V P and Naidu P R 1974Can. J. Chem. 52 788
Reddy K S and Naidu P R 1977Can. J. Chem. 55 76
Timmermans J 1950Physico-chemical constants of pure organic compounds (Amsterdam: Elsevier).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Naidu, G.R., Naidu, P.R. Isentropic compressibilities of ternary systems with 1-alkanol as non-common component. Proc. Indian Acad. Sci. (Chem. Sci.) 91, 393–397 (1982). https://doi.org/10.1007/BF02864174
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02864174