Abstract
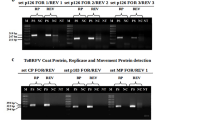
Fifty-nine potato cultivars or breeding clones were planted near Umatilla, OR and/or Pasco, WA, in fields known to be infested with tobacco rattle virus (TRV) and vector nematodes,Paratrichodorus allius Jen. (Sid.). Tubers from these field plots were cut and examined for corky ringspot (CRS) symptoms. Reverse transcription-polymerase chain reaction (RT-PCR) for TRV was conducted on tissue samples from symptomatic and asymptomatic tubers. Sixty-five percent of the symptomatic and 42% of the asymptomatic tissue samples from CRS symptomatic tubers contained detectable TRV. Approximately 2% of plants grown from either symptomatic or asymptomatic tubers contained TRV when tested by ELISA, whereas 20% and 12% of plants grown from symptomatic and asymptomatic tubers, respectively, were positive for TRV by RT-PCR. These results suggest that RT-PCR is a more sensitive assay for detection of TRV. Systemic infections by TRV were detected more often in foliage of CRS-susceptible genotypes. Daughter tubers exhibiting symptoms of CRS, and which contained RT-PCR-detectable TRV, were produced on plants of three genotypes, including one from an asymptomatic parent tuber.
Similar content being viewed by others
Literature Cited
Cadman, C.H. 1959. Potato stem-mottle disease in Scotland. Eur Potato J 2:165–175.
Cadman, C.H. and B.D. Harrison. 1959. Studies on the properties of soil-borne viruses of the tobacco-rattle type occurring in Scotland. Ann Appl Biol 47:542–556.
Converse, R.H. and R.R. Martin. 1990. Elisa methods for plant viruses. Pages 179–196,In: Serological Methods for Detection and Identification of Viral and Bacterial Plant Pathogens. R. Hampton, E. Ball, and S. DeBoer, eds. APS Press, St. Paul, MN.
Crosslin, J.M. and P.E. Thomas. 1995. Detection of tobacco rattle virus in tubers exhibiting symptoms of corky ringspot by polymerase chain reaction. Am Potato J 72:605–609.
Gugerli, P. 1978. The detection of two potato viruses by enzyme-linked immunosorbent assay (ELISA). Phytopath Z 92:51–56.
Harrison, B.D. 1968. Reactions of some old and new British potato cultivars to tobacco rattle virus. Eur Potato J 11:165–176.
Harrison, B.D. and J.I. Cooper. 1974. Studies on the nature of the resistance of potato cultivais to tobacco rattle virus. Pot Res 17:348–349.
Harrison, B.D. and D.J. Robinson. 1982. Genome reconstitution and nucleic acid hybridization as methods of identifying particle-deficient isolates of tobacco rattle virus in potato plants with stem-mottle disease. J Virol Methods 5:255–266.
Harrison, B.D. and D.J. Robinson. 1986. Tobraviruses. Pages 339–369.In: The Plant Viruses, Vol. 2: The Rod-shaped Plant Viruses. M.H.V. van Regenmortel and H. Fraenkel-Conrat, eds. Plenum Press, NY.
Harrison, B.D., D.J. Robinson, W.P. Mowat, and G.H. Duncan. 1983. Comparison of nucleic acid hybridisation and other tests for detecting tobacco rattle virus in narcissus plants and potato tubers. Ann Appl Biol 102:331–338.
Oswald, J.W. and T. Bowman. 1958. Studies on a soil-borne potato virus disease in California. (Abstr.). Phytopathology 48:396.
Ploeg, A.T., D.J.F. Brown, and D.J. Robinson. 1992. Acquisition and subsequent transmission of tobacco rattle virus isolates byParatrichodorus andTrichodorus nematode species. Neth J Plant Path 98:291–300.
Presting, G.G., O.P. Smith, and C.R. Brown. 1995. Resistance to potato leafroll virus in potato plants transformed with the coat protein gene or with vector control constructs. Phytopathology 85:436–442.
Richardson, D.E. 1970. The assessment of varietal reactions to spraing caused by tobacco rattle virus. J Nat Inst Ag Bot 12:112–118.
Richardson, D.E. 1973. An external ringspot of potatoes. PI Path 22:120–122.
Samson, R.G., T.C. Allen, and J.L. Whitworth. 1993. Evaluation of direct tissue blotting to detect potato viruses. Am Potato J 70:257–265.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Crosslin, J.M., Thomas, P.E. & Brown, C.R. Distribution of tobacco rattle virus in tubers of resistant and susceptible potatoes and systemic movement of virus into daughter plants. Am. J. Pot Res 76, 191–197 (1999). https://doi.org/10.1007/BF02854221
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02854221