Abstract
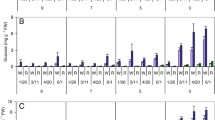
Mechanical handling of Norchip (cv.) potatoes resulted in sucrose concentrations that exceeded 1% (fresh wt basis) 10 days after treatment. More than 65% of the maximal sugar accumulation occurred within 5 days of handling. Potatoes increased in their ability to produce sucrose as storagetime increased. Sucrose accumulation in response to handling stress was not mediated via gross physical disruption of the amyloplast membrane. Electron micrographs indicated that starch granules of both handled and control potatoes were surrounded by intact and continuous plastid (amyloplast) membranes throughout the major portion (1 to 8 months) of the storage season (8.9 C). Glucose levels did not increase at a similar rate compared to changes in sucrose concentration. Results suggest that the majority of the sucrose, which occurred during storage and/or handling stress, was not efficiently converted to reducing sugars via the enzyme invertase.
Sucrose and glucose concentrations in all potatoes began to increase and chip color darkened as tubers senesced (9 to 10 months storage). Examination of electron micrographs during this storage period revealed that the bi-layers of the amyloplast membrane were separating. The mechanical handling of senescing potatoes (10 months in storage) resulted in a shift of the plastid membrane to a more disrupted state over that noted in the control.
The variation in chip color induced by handling and/or storage time in Norchip potatoes was closely associated with the variation in glucose content (r = 0.953).
Resumen
Las micrografías electrónicas han indicado que los gránulos de almidón, tanto de los tubérculos de papa manipulados como de aquellos en el testigo, se encuentran circundados por las membranas de los plástidos (amiloplastos), intactas y contínuas, a través de la mayor parte (1 a 8 meses) de la temporada de almacenamiento (8.9 C). La sucrosa aumentó marcádamente cuando papas del cultivar Norchip fueron expuestas al expuestas al estrés a la manipulación. El manipuleo mecánico provocó que los valores de sucrosa excedieran el 1% (en base a peso fresco) 10 días después del tratamiento. Más del 65% de la máxima acumulación de azúcar ocurrió dentro de 5 días de manipulación. Conforme los tubérculos envejecen en el almacenamiento, su capacidad para producir sucrosa, en respuesta al estrés a la manipulación, aumenta. El mecanismo por el cual la sucrosa aumenta, debido a la manipulación no fue mediatizado por la fuerte desorganización física de la membrana de los amiloplastos. El incremento en la concentración de sucrosa no estuvo acompañado por un aumento proporcional en glucosa. Los resultados sugieren que la mayor parte de la sucrosa que se produce a continuación del envejecimiento, y lo estrés al manipuleo, queda aislado es un compartimiento celular protegido de la acción de la invertasa.
Las concentraciones de sucrosa y glucosa en los tubérculos no manipulados empezaron a aumentar, y el color de las papas fritas a la inglesa a obscurecer, conforme empezó el envejecimiento de los tubérculos (9 a 10 meses de almacenamiento). El examen de la micrografías electrónicas durante este período de envejecimiento reveló que las 2 capas de las membranas de los amiloplastos estaban separándose. El manipuleo de los tubérculos que ya presentaban endulzamiento por envejecimiento provocó que las membranas de los amiloplastos aceleran su tasa de desorganización. La variación en color, de las papas fritas a la inglesa, inducida por el manipuleo y/o el estrés al envejecimiento, en las papas Norchip, estuvo asociada, principalmente, con la variación en el contenido de glucosa (r = 0,953).
Similar content being viewed by others
Literature Cited
ap Rees, T., W.L. Dixon, C.L. Pollock and F. Franks. 1981. Low temperature sweetening of higher plants.In: J. Friend and M.J.C. Rhodes (eds.). Recent advances in the biochemistry of fruits and vegetables. Academic Press, London, New York. pp. 41–61.
Ewing, E.E., A.H. Senesac and J.B. Sieczka. 1981. Effects of short periods of chilling and warming on potato sugar content and chipping quality. Am Potato J 58:633–647.
Iritani, W.M. and L. Weller. 1978. Influence of low fertility and vine killing on sugar development in apical and basal portions of Russet Burbank potatoes. Am Potato J 55:239–246.
Isherwood, F.A. 1976. Mechanism of starch-sugar interconversion inSolanum tuberosum. Phytochemistry 15:33–41.
Lulai, E.C. and P.H. Orr. 1980. Quality-testing facilities for grower use at the potato research laboratory. Am Potato J 57:622–628.
Lulai, E.C., J.R. Sowokinos and J.A. Knoper. 1986. Translucent tissue defects inSolanum tuberosum L. II. Alterations in lipolytic acyl hydrolase, lipoxygenase, and morphology of mitochondria and amyloplasts. Plant Physiol 80:424–428.
Mares, D.J., J.R. Sowokinos and J.S. Hawker. 1985. Carbohydrate metabolism in developing potato tubers.In: P.H. Li (ed.). Potato Physiology. Academic Press, New York. pp. 279–327.
Ohad, I., I. Friedberg, Z. Ne’eman and M. Schramm. 1971. Biogenesis and degradation of starch I. The fate of the amyloplast membrane during maturation and storage of potato tubers. Plant Physiol 47:465–477.
Orr, P.H., J.R. Sowokinos and J.L. Varns. 1985. Sugar changes and chipping responses of Norchip tubers after handling from storage. Amer Soc of Agr Engr Paper No. 85-6021.
Owings, T.R., W.M. Iritani and C.W. Nagel. 1978. Respiration rates and sugar accumulation in normal and moisture stressed Russet Burbank potatoes. Am Potato J 55:211–220.
Pressey, R. 1969. Role of invertase in the accumulation of sugars in cold-stored potatoes. Am Potato J 46:291–297.
Pressey, R. and R. Shaw. 1966. Effect of temperature on invertase, invertase inhibitor, and sugars in potato tubers. Plant Physiol 41:1657–1661.
Shallenberger, R.S., O. Smith and R.H. Treadway. 1959. Role of the sugars in the browning reaction in potato chips. J Agric Food Chem 7:274–277.
Sieczka, J.B. and C. Maatta. 1986. The effects of handling on chip color and sugar content of potato tubers. Am Potato J 63:363–372.
Smith, O. 1968. Mineral nutrition of the potato.In: O. Smith (ed.), Potatoes: production, storage, processing. AVI Publishing Company, Inc., Westport, Connecticut. pp. 183–241.
Sowokinos, J.R., E.C. Lulai and J.A. Knoper. 1985. Translucent tissue defects inSolanum tuberosum L. I. Alterations in amyloplast membrane integrity, enzyme activities, sugars and starch content. Plant Physiol 78:489–494.
Spurr, A.R. 1969. A low-viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct Res 26:31–43.
Timm, H., M. Yamaguchi, M.D. Clegg and J.C. Bishop. 1968. Influence of high-temperature exposure on sugar content and chipping quality of potatoes. Am Potato J 45:359–365.
Wetstein, H.Y. and C. Sterling. 1978. Integrity of amyloplast membranes in stored potato tubers. Z. Pflanzenphysiol 90:373–378.
Williams, M.G. and E.K. Adrian. 1977. The use of elemental iodine to enhance staining of thin sections to be viewed in the electron microscope. Stain Technol 52:269–272.
Author information
Authors and Affiliations
Additional information
Agricultural Experiment Station, University of Minnesota Scientific Journal Series No. 15, 155.
A laboratory cooperatively operated by the USDA-ARS; The Minnesota Agricultural Experiment Station; The North Dakota Agricultural Experiment Station and the Red River Valley Potato Grower’s Association.
Rights and permissions
About this article
Cite this article
Sowokinos, J.R., Orr, P.H., Knoper, J.A. et al. Influence of potato storage and handling stress on sugars, chip quality and integrity of the starch (amyloplast) membrane. American Potato Journal 64, 213–226 (1987). https://doi.org/10.1007/BF02853559
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02853559