Abstract
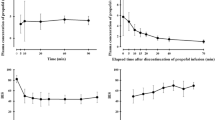
This study compared the ability of the physiology-based pharmacokinetic (PBPK) model with that of compartmental models used in propofol infusion devices to predict the pharmacokinetics and pharmacodynamics of propofol in various patient groups (children, pregnant women, young men, normal weight adults, and obese adults). With a PBPK model, loss of consciousness (LOC) and recovery of consciousness (ROC) corresponded to a narrow range of brain tissue concentrations (2.2–4.0 mg/L). With the compartmental models, predicted effect concentrations were also within a narrow range at LOC, but were outside the range at ROC. In individuals of normal weight, coefficients of variation (CV) of the predicted brain or effect concentrations at LOC were in a similar range—between 18% and 32%. In obese individuals, however, interindividual CV values for brain or effect concentrations were 41% (PBPK) and 93% (compartmental). This comparison suggests the increased flexibility of PBPK models over compartmental models, the latter of which rely heavily on the patient group from which the model was derived. The incorporation of PBPK models may provide target-controlled infusions with enhanced ability to predict response in a wide variety of patients.
Similar content being viewed by others
References
Gepts E, Camu F, Cockshott ID, Douglas EJ. Disposition of propofol administered as constant rate intravenous infusions in humans.Anesth Analg. 1987; 66: 1256–1263.
Marsh B, White M, Morton N, Kenny GN. Pharmacokinetic model driven infusion of propofol in children.Br J Anaesth. 1991; 67: 41–48.
Munoz HR, Cortinez LI, Ibacache ME, Altmann C. Estimation of the plasma effect site equilibration rate constant (keo) of propofol in children using the time to peak effect.Anesthesiology. 2004; 101: 1269–1274.
Kazama T, Ikeda K, Morita K, et al. Investigation of effective anesthesia induction doses using a wide range of infusion rates with undiluted and diluted propofol.Anesthesiology. 2000; 92: 1017–1028.
Absalom A, Kenny G. ‘Paedfusor’ pharmacokinetic data set.Br J Anaesth. 2005; 95: 110.
Kazama T, Morita K, Ikeda T, Kurita T, Sato S. Comparison of predicted induction dose with predetermined physiologic characteristics of patients and with pharmacokinetic models incorporating those characteristics as covariates.Anesthesiology. 2003; 98: 299–305.
Ginsberg G, Hattis D, Russ A, Sonawane B. Physiologically based pharmacokinetic (PBPK) modeling of caffeine and theophylline in neonates and adults: implications for assessing children’s risks from environmental agents.J Toxicol Environ Health. 2004; 67: 297–329.
Theil FP, Guentert TW, Haddad S, Poulin P. Utility of physiologically based pharmacokinetic models to drug development and rational drug discovery candidate selection.Toxicol Lett. 2003; 138: 29–49.
Rowland M, Balant L, Peck C. Physiologically based pharmacokinetics in drug development and regulatory science: a workshop report (Georgetown University, Washington, DC, May 29–30, 2002).AAPS Pharm Sci. 2004; 6: 1–12.
Valtonen M, Iisalo E, Kanto J, Rosenberg P. Propofol as an induction agent in children: pain on injection and pharmacokinetics.Acta Anaesthesiol Scand. 1989; 33: 152–155.
Raoof AA, Van Obbergh LJ, Verbeeck RK. Propofol pharmacokinetics in children with biliary atresia.Br J Anaesth. 1995; 74: 46–49.
Saint-Maurice C, Cockshott ID, Douglas EJ, Richard MO, Harmey JL. Pharmacokinetics of propofol in young children after a single dose.Br J Anaesth. 1989; 63: 667–670.
Kanto J, Rosenberg P. Propofol in cesarean section. A pharmacokinetic and pharmacodynamic study.Methods Find Exp Clin Pharmacol. 1990; 12: 707–711.
Mertens MJ, Olofsen E, Burm AG, Bovill JG, Vuyk J. Mixed-effects modeling of the influence of alfentanil on propofol pharmacokinetics.Anesthesiology. 2004; 100: 795–805.
Ickx B, Cockshott ID, Barvais L, et al. Propofol infusion for induction and maintenance of anaesthesia in patients with end-stage renal disease.Br J Anaesth. 1998; 81: 854–860.
Lysakowski C, Dumont L, Pellegrini M, Clergue F, Tassonyi E. Effects of fentanyl, alfentanil, remifentanil and sufentanil on loss of consciousness and bispectral index during propofol induction of anesthesia.Br J Anaesth. 2001; 86: 523–527.
Willmann S, Schmitt W, Keldenich J, Lippert J, Dressman JB. A physiological model for the estimation of the fraction dose absorbed in humans.J Med Chem. 2004; 47: 4022–4031.
Willmann S, Lippert J, Sevestre M, Solodenko J, Fois F, Schmitt W. PK-Sim: a physiologically based pharmacokinetic ‘whole-body’ model.Biosilico. 2003; 1: 121–124.
Blackburn ST.Maternal, Fetal, & Neonatal Physiology: A Clinical Perspective. 2nd ed. Philadelphia, Pa: Elsevier Publishing; 2003.
Hardman JG, Limbird LE.Goodman and Gilman’s: The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw Hill; 2001.
Jones RDM, Chan K, Andrew LJ. Pharmacokinetics of propofol in children.Br J Anaesth. 1990; 65: 661–667.
Murat I, Billard V, Vernois J, et al. Pharmacokinetics of propofol after a single dose in children aged 1–3 years with minor burns. Comparison of three data analysis approaches.Anesthesiology. 1996; 84: 526–532.
Knibbe CA, Melenhorst-de Jong G, Mestrom M, et al. Pharmacokinetics and effects of propofol 6% for short-term sedation in paediatric patients following cardiac surgery.Br J Clin Pharmacol. 2002; 54: 415–422.
Ludbrook GL, Visco E, Lam AM. Relation between brain concentrations, electroencephalogram, middle cerebral artery blood flow velocity, and cerebral oxygen extraction during induction of anesthesia.Anesthesiology. 2002; 97: 1363–1370.
Mi WD, Sakai T, Kudo M, Matsuki A. Performance of bispectral index and auditory evoked potential monitors in detecting loss of consciousness during anaesthetic induction with propofol with and without fentanyl.Eur J Anaesth. 2004; 10: 807–811.
Flaishon R, Windsor A, Sigl J, Sebel PS. Recovery of consciousness after thiopental or propofol: bispectral index and the isolated forearm technique.Anesthesiology. 1997; 86: 613–619.
Servin F, Farinotti R, Haberer J, Desmonts J. Propofol infusion for maintenance of anesthesia in morbidly obese patients receiving nitrous oxide.Anesthesiology. 1993; 78: 657–665.
Levitt DG, Schnider TW. Human physiologically based pharmacokinetic model for propofol.BMC Anaesthesiol. 2005; 5: 1–4.
Frohlich MA, Dennis DM, Shuster JA, Melker RJ. Precision and bias of target controlled propofol infusion for sedation.Br J Anaesth. 2005; 94: 434–437.
Bouillon T, Shafer SL. Does size matter?Anesthesiology. 1998; 89: 557–560.
Slepchenko G, Simon N, Goubaux B, et al. Performance of target-controlled sufentanil infusion in obese patients.Anesthesiology. 2003; 98: 65–73.
Absalom A, Amutike D, Lal A, White M, Kenny GNC. Accuracy of the ‘Paedfusor’ in children undergoing cardiac surgery or catheterization.Br J Anaesth. 2003; 91: 507–513.
Kataria BK, Ved SA, Nicodemus HF, et al. The pharmacokinetics of propofol in children using three different data analysis approaches.Anesthesiology. 1994; 80: 104–122.
Wintermark M, Lepori D, Cotting J, et al. Brain perfusion in children: evolution with age assessed by quantitative perfusion computed tomography.Pediatrics. 2004; 113: 1642–1652.
Casati A, Putzu M. Anesthesia in the obese patient: pharmacokinetic considerations.Br J Anaesth. 2005; 17: 134–145.
Rigby-Jones AE, Nolan JA, Priston MJ, Wright P, Sneyd R, Wolf AR. Pharmacokinetics of propofol infusions in critically ill neonates, infants, and children in an intensive care unit.Anesthesiology. 2002; 97: 1393–1400.
Gan TJ, Glass PS, Sigl J, et al. Women emerge from general anesthesia with propofol/alfentanil/ nitrous oxide faster than men.Anesthesiology. 1999; 90: 1283–1287.
Milne SE, Kenny GN, Schraag S. Propofol sparing effect of remifentanil using closed-loop anaesthesia.Br J Anaesth. 2003; 90: 623–629.
Schnider TW, Minto CF, Gambus PL, et al. The influence of method of administration and covariates on the pharmacokinetics of propofol in adult volunteers.Anesthesiology. 1998; 88: 1170–1182.
Vuyk J, Engbers FHM, Burm AG, Vletter A, Bovill JG. Performance of computer-controlled infusion of propofol: an evaluation of five pharmacokinetic parameter sets.Anesthesiology. 1995; 81: 1275–1282.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Edginton, A.N., Schmitt, W. & Willmann, S. Application of physiology-based pharmacokinetic and pharmacodynamic modeling to individualized target-controlled propofol infusions. Adv Therapy 23, 143–158 (2006). https://doi.org/10.1007/BF02850355
Issue Date:
DOI: https://doi.org/10.1007/BF02850355