Abstract
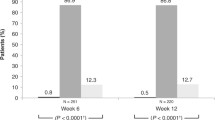
In an open-label 12-week study, the safety and efficacy of bimatoprost 0.03% was evaluated in 55 patients with open-angle glaucoma or ocular hypertension inadequately controlled by topical beta-blocker monotherapy. Patients discontinued their topical beta-blocker therapy at the baseline visit and began bimatoprost monotherapy that evening. Study visits were at 6 and 12 weeks postbaseline. Bimatoprost reduced intraocular pressure (IOP) 4.5 mm Hg (21.5%; P< .001) from baseline at week 6 and 4.2 mm Hg (19.6%; P< .001) at week 12. Patients were more likely to achieve low target pressures with bimatoprost than with topical beta-blockers. Conjunctival hyperemia was the most commonly reported adverse event. The findings from this study indicate bimatoprost monotherapy provides a substantially greater IOP reduction than topical beta-blocker therapy and allows more patients to achieve a low target pressure. Bimatoprost is an effective alternative to topical beta-blockers for the treatment of glaucoma and ocular hypertension.
Similar content being viewed by others
References
Jonsson B, Krieglstein G, Kobelt G, eds. Primary open-angle glaucoma: differences. In:International Treatment Patterns and Cost. London, England: Taylor & Francis, LTD; 1998.
Heijl A, Leske C, Bengtsson B, et al. Reduction of intraocular pressure and glaucoma progression. Results from the Early Manifest Glaucoma Trial.Arch Ophthalmol. 2002;120:1268–1279.
Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma.Arch Ophthalmol. 2002;120:701–713.
The AGIS Investigators. The advanced glaucoma intervention study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration.Am J Ophthalmol. 2000;130: 429–440.
Brandt JD, VanDenburgh AM, Chen K, Whitcup SM, for the Bimatoprost Study Group I. Comparison of once-or twice-daily bimatoprost with twice-daily timolol in patients with elevated IOP: a 3-month clinical trial.Ophthalmology. 2001;108:1023–1031; discussion 1032.
Higginbotham EJ, Schuman JS, Goldberg I, et al. One-year randomized study comparing bimatoprost and timolol in glaucoma and ocular hypertension.Arch Ophthalmol. 2002;120:1286–1293.
Zimmerman T. Medicinal therapy for glaucoma. In: Eid TM, Spaeth GL, eds.The Glaucomas: Concepts and Fundamentals. Philadelphia, Pa: Lippincott Williams & Wilkins; 2000:227–245.
McMahon CD, Shaffer RN, Hoskins HD Jr, Hetherington J Jr. Adverse effects experienced by patients taking timolol.Am J Ophthalmol. 1979;88:736–738.
Noecker RJ, Dirks MS, Choplin NT, et al. A six-month randomized clinical trial comparing the IOP-lowering efficacy of bimatoprost and latanoprost in patients with ocular hypertension or glaucoma.Am J Ophthalmol. 2003;135:55–63.
Netland PA, Landry T, Sullivan EK, et al, and the Travoprost Study Group. Travoprost compared with latanoprost and timolol in patients with open-angle glaucoma or ocular hypertension.Am J Ophthalmol. 2001;132:472–484.
Singh K, Spaeth G, Zimmerman T, Minckler D. Target pressure-glaucomatologists’ holey grail.Ophthalmology. 2000;107:629–630.
Asrani S, Zeimer R, Wilensky J, Gieser D, Vitale S, Lindenmuth K. Large diurnal fluctuations in intraocular pressure are an independent risk factor in patients with glaucoma.J Glaucoma. 2000;9:134–142.
Pillunat LE, Larsson LI; European and Canadian Latanoprost Study Group. Intraocular pressure after replacement of current dual therapy with latanoprost monotherapy in patients with open angle glaucoma.Br J Ophthalmol. 2003;87:1492–1496.
Dubiner HB, Sircy MD, Landry T, et al. Comparison of the diurnal ocular hypotensive efficacy of travoprost and latanoprost over a 44-hour period in patients with elevated intraocular pressure.Clin Ther. 2004;26:84–91.
Choplin N, Bernstein P, Batoosingh AL, Whitcup SM; Bimatoprost/Latanoprost Study Group. A randomized, investigator-masked comparison of diurnal responder rates with bimatoprost and latanoprost in the lowering of intraocular pressure.Surv Ophthalmol. 2004;49(suppl 1):S19-S25.
Enright PL, McClelland RL, Newman AB, Gottlieb DJ, Lebowitz MD. Underdiagnosis and under-treatment of asthma in the elderly. Cardiovascular Health Study Research Group.Chest. 1999;116: 603–613.
Lama PJ. Systemic adverse effects of beta-adrenergic blockers: an evidence-based assessment.Am J Ophthalmol. 2002;134:749–760.
Valuck RJ, Perlman JI, Anderson C, Wortman GI. Co-prescribing of medications used to treat obstructive lung disease, congestive heart failure and depression among users of topical beta-blockers: estimates from three US Veterans Affairs Medical Centers.Pharmacoepidemiol Drug Saf. 2001;10:511–516.
Sherwood M, Brandt J, for the Bimatoprost Study Groups 1 and 2. Six-month comparison of bimatoprost once-daily and twice-daily with timolol twice-daily in patients with elevated intraocular pressure.Surv Ophthalmol. 2001;45 (suppl 4):S361-S368.
Schuman JS. Effects of systemic beta-blocker therapy on the efficacy and safety of topical brimonidine and timolol. Brimonidine Study Groups 1 and 2.Ophthalmology. 2000;107:1171–1177.
Steinert RF, Thomas JV, Boger WP 3rd. Long-term drift and continued efficacy after multiyear timolol therapy.Arch Ophthalmol. 1981;99:100–103.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Quinones, R., Earl, M.L. The efficacy of bimatoprost 0.03% monotherapy in patients previously using topical beta-blocker monotherapy for the treatment of glaucoma or ocular hypertension. Adv Therapy 21, 370–379 (2004). https://doi.org/10.1007/BF02850101
Issue Date:
DOI: https://doi.org/10.1007/BF02850101