Abstract
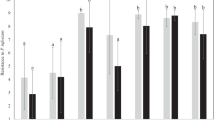
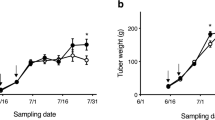
Blackspot pigment development was measured by three methods: 1) impact, 2) abrasive peeling, and 3) oxidation of tuber homogenates. Genotypes and storage periods were used as variable treatments. All methods were able to separate genotypes based on blackspot resistance although the abrasive peel and homogenization methods provided more consistent results than the impact bruise method. Bruise resistant genotypes transferred their resistance to progeny in crosses with a susceptible genotype which was correlated with tyrosine content. According to the traditional impact bruise technique, tuber resistance to blackspot increased during the eight-to ninemonth storage at 3 C. In contrast, resistance was found to decrease with the homogenization procedure and did not change with the abrasive peel technique. Free tyrosine was consistently associated with blackspot pigment development but chlorogenic acid and caffeic acid were not.
Compendio
Se midió el desarrollo del pigmento de la mancha negra por tres métodos: 1) impacto, 2) pelado con abrasivos y 3) oxidation de homogenados de tubérculo. Se utilizaron a los genotipos y periodos de almacenamiento como tratamiento variables. Todos los métodos permitieron separar a los genotipos basándose en la resistencia a la mancha negra aunque el pelado con abrasivos y los métodos de homogenización proveyeron resultados más consistentes que el método de magulladuras al impacto. Los genotipos resistentes a las magulladuras transfirieron esta resistencia a sus progenies en cruzamientos con un genotipo susceptible que estaba correlacionado con el contenido de tiroxina. De acuerdo con la técnica tradicional de magulladuras al impacto, la resistencia de los tubérculos a la mancha negra se incrementó durante los ocho a nueve meses de almacenamiento a 3 C. Por el contrario, la resistencia disminuyó con el procedimiento de homogenización y se mantuvo estable con la técnica de pelado con abrasivos. La tiroxina libre estuvo consistentemente asociada con el desarrollo de la pigmentation de la mancha negra, pero los ácidos clorogénico y cafeínico no mostraron tal asociación.
Similar content being viewed by others
Literature Cited
Bidlingmeyer, B.A., S.A. Cohen and T.L. Tarvon. 1984. Rapid analysis of amino acids using pre-column derivitization. Journal of Chromatography 336:93–104.
Boyd, A.E.W. and G.R. White. 1956. Effect of potash on the internal blackening of potatoes. Plant Pathology 5:107–109.
Kunkel, R. and W.H. Gardner. 1958. Blackspot of Russet Burbank potatoes. American Society of Horticultural Science 73:436–444.
McDole, R.E., D.T. Westerman, G.D. Kleinschmidt, G.E. Kleinkopf and J. Ogala. 1987. Idaho fertilizer guide for potatoes. Current Information Series Number 261. University of Idaho, Moscow, ID.
Mondy, N.I., E. Owens Mobley and S. Bond Gedde-Dahl. 1967. Influence of potassium fertilization on enzymatic activity, phenolic content and discoloration of potatoes. Journal of Food Science 32:378–381.
Mondy, N.I., B.P. Klein and L.I. Smith. 1960. The effect of maturity and storage on phenolic content, enzymatic activity and discoloration of potatoes. Food Research 25:693–705.
Pavek, J.J., D. Corsini and F. Nissley. 1985. A rapid method for determining blackspot susceptibility of potato clones. Am Potato J 62:511–517.
Pavek, J., C.R. Brown, M.W. Martin, and D.L. Corsini. (in press). Inheritance of blackspot bruise resistance in potato. Am Potato J.
Sapers, G.W., F.W. Douglas, Jr., A. Bilyk, A.-F. Hsu, H.W. Dower, L. Garzarella and M. Kozempel. 1989. Enzymatic browning in Atlantic potatoes and related cultivars. Journal of Food Science 54:362–365.
Shippers, P.A. 1971. Measurement of blackspot susceptibility of potatoes. Am Potato J 43:71–81.
Stark, J.C., D.L. Corsini, P.J. Hurley and R.B. Dwelle. 1985. Biochemical characteristics of potato clones differing in blackspot susceptibility. Am Potato J 62:657–666.
Umaerus, M. and K. Olsson. 1974. Varietal differences in tyrosine and chlorogenic acid in relation to enzymic discoloration of potato tubers. Potato Research 17:157–158.
Vertregt, N. 1968. Relation between blackspot and composition of the potato tuber. Eur Potato J 11:34–44.
Weaver, M.L., E. Hautala and R.M. Reeve. 1970. Distribution of oxidase enzymes in potato tubers relative to blackspot susceptibility. I. Phenolases. Am Potato J 47:479–488.
Weaver, M.L., H.A. Steen and K.N. Nilsen. 1966. Quantitative measurement of discoloration pigment in injured potato tissue by densitometry. Am Potato J 43:412–417.
Author information
Authors and Affiliations
Additional information
Hort/LA Paper No. 92-03. Project No. 0766. College of Agriculture and Home Economics Research Center, Washington State University, Pullman, WA 99164.
Rights and permissions
About this article
Cite this article
Dean, B.B., Jackowiak, N., Nagle, M. et al. Blackspot pigment development of resistant and susceptibleSolanum Tuberosum L. genotypes at harvest and during storage measured by three methods of evaluation. American Potato Journal 70, 201–217 (1993). https://doi.org/10.1007/BF02849309
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02849309