Abstract
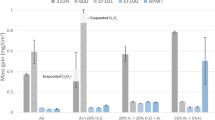
The ignition-burn conditions of several ferrous metals, including AISI1025, AISI 4140, ductile iron and 304, 410, 17-4 PH, and CA15 stainless steels have been determined. The oxygen conditions involved velocities of approximately 430 meters per second (1400 feet per second) and pressures of 20 to 68 atm (300 to 1000 psig). When 1025 steel specimens were fractured in 68 atm (1000 psig), 427 mps (1400 fps) oxygen, this steel was found to ignite and burn at a nominal temperature of about 480 K (400 °F). Under similar conditions, AISI 4140 ignited at about 590 K (600 °F) and ductile cast iron ignited at a temperature of (530 K (200 °F) or less. Stainless steels (304, 17-4 PH, and CA15) ignited at about 590 K (600 °F) and 410 stainless steel, which appeared more resistant to ignition, was found to ignite at about 645 K (700 °F). The ignition temperature was observed to increase for all materials as the oxygen pressure was reduced.
Similar content being viewed by others
References
Oxygen Compressors and Pumps Symposium, Atlanta, Georgia, Nov. 9–11, 1971, Compressed Gas Association, Inc.
M. D. Hersey:American Society of Naval Engineers, 1924, Vol. 36, pp. 231–243.
W. C. Reynolds: NASA Tech. Note TND-182, October 1959.
Paul R. Hill, David Adamson, Douglas H. Foland, and Walter E. Bressette: National Advisory Committee for Aeronautics Research Memorandum, NACA RM L55L23b.
L. E. Dean and W. R. Thompson:ARS Journal, July 1961, pp. 917–923.
A. H. Tench, Hans M. Roder, and A. F. Clark:Combustion of Metals in Oxygen—Phase II: Bulk Burning Experiments, NBSIR 73:345, December 1973.
L. Kirschfeld:Archiv Eisenhuttenw., September 1962, Vol. 33, pp. 617–621.
L. Kirschfeld:Archiv Eisenhuttenw., July 1968, Vol. 39, pp. 535–539.
L. Kirschfeld:Archiv Eisenhuttenw., July 1968, Vol. 39, pp. 823–826.
L. Kirschfeld:Angew Chem., 1959, Vol. 71, pp. 663–667.
L. Kirschfeld:Metall, February 1967, Vol. 21, pp. 98–102.
L. Kirschfeld:Metall, 1960, Vol. 14, pp. 213–219.
L. Kirschfeld:Metall, August 1960, pp. 792–796.
L. Kirschfeld:Archiv Eisenhuttenw., January 1961, Vol. 32, pp. 57–62.
L. Kirschfeld:Metall, September 1961, Vol. 15, pp. 873–878.
P. L. Harrison and A. D. Yoffe:Proceedings of Royal Society, Vol. 261A, pp. 537–370.
G. J. Nihart and C. P. Smith: AMRL-TDR-64-76, Union Carbide Corp., Linde Div., October 1964.
W. A. Riehl, C. F. Key, and J. B. Gayle: NASA Tech. Report R-180.
“Reactivity of Metals with Liquid and Gaseous Oxygen,” DMIC, Battelle Memorandum 163, January 15, 1963.
Lewis Greenspan:Review of Scientific Instruments, February 1958, Vol. 29, pp. 172–173.
Robert J. Schwinghamer and Carlo F. Key: NASA TMX-64899, November 29, 1974.
C. F. Key, J. C. Austin, and J. W. Bransford: NASA TMX-64783, September 1973.
D. L. Pippen and J. S. Stradling:Material Research and Standards, June 1971, Vol. 53, pp. 35–53.
C. F. Key:Materials Research and Standards, June 1971, Vol. 11, pp. 28–51.
C. F. Key:Compatibility of Materials with LOX—Vol. I, NASA TMX-64711, October 1972.
W. R. Blackstone, B. B. Baber, and P. M. Ku: Southwest Research Institute, AFAPL TR-67-41, December 1967.
R. J. Schwinghamer: NASA TMX-64634, January 4, 1972.
Jack L. Christian, James E. Chafey, Abraham Hurlich, James F. Watson, and William E. Witzell:Metal Progress, April 1963, pp. 100–103, 122–123.
A. A. Wells:British Welding Journal, September 1955, pp. 392–400.
H. Bauer, W. Wegener, and K. F. Windgassen:Cryogenics, June 1970, Vol. 10, pp. 241–248.
H. Bauer, W. Wegener, and K. F. Windgassen:Cryogenics, December 1971, Vol. 11, pp. 469–476.
F. E. Littman, F. M. Church, and E. M. Kinderman:Journal of Less Common Metals, 1961, Vol. 3, pp. 367–378.
I. Glassman et al: NATO AGARD Annual Meeting, February 1970, Conference Proceedings No. 52, pp. 19 (1–30).
O. Kubaschewski and E. Evans:Metallurgical Thermochemistry, Pergamon Press, Oxford, 1958.
K. K. Kelley: Bulletin 584, Bureau of Mines, 1960.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bates, C.E., Wren, J.E., Monroe, R. et al. Ignition and combustion of ferrous metals in high pressure, high velocity, gaseous oxygen. JMES 1, 61–76 (1979). https://doi.org/10.1007/BF02833991
Issue Date:
DOI: https://doi.org/10.1007/BF02833991