Abstract
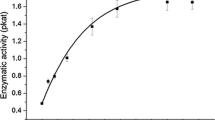
An optical biosensor with a stirred cuvette has been used to monitor the interaction of immobilized wheat germ agglutinin (WGA) with two water-soluble cationic porphyrins. The association constants (K a) of the free base porphyrin and its Zn(II) complex form were 2.66 and 27.31×105 L/mol at 20°C respectively. The interactions of the free base porphyrin were further investigated at temperatures between 15°C and 37°C. The thermodynamics parameters, changes in free energy, enthalpy and entropy, were −31.23, 22.92, 54.15 kJ/mol respectively. The heat capacity change was −355.53 J·mol−1·K−1. The binding was driven by entropic contribution, and showed strong enthalpy-entropy compensation. It was governed primarily by hydrophobic forces.
Similar content being viewed by others
References
Bonnett R. Photosensitizers of the Porphyrin and Phtalocyanine Series for Photodynamic Therapy [J].Chem Soc Rev, 1995,24: 19–33.
Dougherty T J, Gomer C J, Henderson B W,et al. Photodynamic Therapy [J].J Natl Cancer Inst, 1998,90(12): 889–905.
Wang Lei, Zhu Xunjin, Fu Shitao,et al. Synthesis of Water-Soluble Metalloporphyrins and Their Inhibiting Action on the Growth Metabolism ofE. coli [J].Chem J Chin Univ, 2003,24(7): 1145–1149 (Ch).
Tan A M, Xu B, Huang S Q,et al. Thermochemical Study on the Growth Metabolism of Human Promyelocytic Leukemia HL-60 Cells Inhibited by Water-Soluble Metalloporphyrins [J].Thermochim Acta, 1999,333: 99–102.
Kreimer-Birnbaum M. Modified Porphyrins, Chlorins, Phthalocyanines, and Purpurins: Second-Generation Photosensitizers for Photodynamic Therapy [J].Semin Hematol, 1989,26(2): 157–173.
Wirth M, Fuchs A, Wolf M,et al. Lectin-Mediated Drug Targeting: Preparation, Binding Characteristics, and Antiproliferative Activity of Wheat Germ Agglutinin Conjugated Doxorubicin on Caco-2 Cells [J].Pharm Res, 1998,15(7): 1031–1037.
Gabor F, Kausegger U, Wrth M. The Interaction between Wheat Germ Agglutinin and Other Plant Lectins with Prostate Cancer Cells Du-145 [J].Int J Pharm, 2001,221(1–2): 35–47.
Kenoth R, Reddy D R, Maiya B G,et al. Thermodynamic and Kinetic Analysis of Porphyrin Binding to Trichosanthes Cucumerina Seed Lectin [J].Eur J Biochem, 2001,268 (21): 5541–5549.
Sultan N A, Maiya B G, Swamy M J. Thermodynamic Analysis of Porphyrin Binding to Momordica Charantia (Bitter Gourd) Lectin [J].Eur J Biochem, 2004,271(15): 3274–3282.
Leatherbarrow R J, Edwards P R. Analysis of Molecular Recognition Using Optical Biosensors [J].Cur Opin Chem Boil, 1999,3(5): 544–547.
Zeng X X, Nakaaki Y, Murata T,et al. Chemoenzymatic Synthesis of Glycopolypeptides Carrying Alpha-Neu5Ac (2→3)-beta-D-Gal-(1→3)-alpha-D-GalNAc, Beta-D-Gal-(1→3)-alpha-D-GalNAc, and Related Compounds and Analysis of Their Specific Interactions with Lectins [J].Arch Biochem Biophys, 2000,383(1): 28–37.
Davila J, Harriman A. Photoreactions of Macrocyclic Dyes Bound to Human Serum Albumin [J].Photochem Photobiol, 1990,51(1): 9–19.
Komath S S, Kenoth R, Giribabu L,et al. Fluorescence and Absorption Spectroscopic Studies on the Interaction of Porphyrins with Snake Gourd (Trichosanthes anguina) Seed Lectin [J].J Photochem Photobiol B: Biol, 2000,55(1): 49–55.
Ross P D, Subramanian S. Thermodynamics of Protein Association Reactions. Forces Contributing to Stability [J].Biochem, 1981,20(11): 3096–3102.
Sturtevant J M. Heat Capacity and Entropy Changes in Processes Involving Proteins [J].Proc Natl Acad Sci USA, 1977,74: 2236–2240.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: Supported by the National Natural Science Foundation of China (30370366)
Biography: QIN Yimin (1976-), female, Ph. D., research direction: applied biochemistry and molecular biology.
Rights and permissions
About this article
Cite this article
Yimin, Q., Nina, P. & Guolin, Z. Application of an optical biosensor to study the interaction between porphyrins and wheat germ agglutinin. Wuhan Univ. J. Nat. Sci. 11, 432–436 (2006). https://doi.org/10.1007/BF02832138
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02832138