Abstract
Objective
To investigate the relationship between P120ctn translocation and hepatocellular carcinoma cell malignant features and the relationship between p 120ctn and β-catenin translocation in cell signaling.
Methods
Human hepatocellular carcinoma cells were over expressed with p 120ctn isoform 3A using a DNA transfection method. The effects of transfection and expression of p 120{ctn} and its binding capacity to E-cadherin were examined using immunoprecipitation and immunoblotting methods. p 120ctn subcellular localization and its relation with β-catenin were detected using immunofluorescent microscopy. p 120{ctn} phosphorylation was produced by EGF treatment. Cell adhesion, cell migration and cell proliferation were also examined in this study.
Results
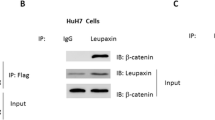
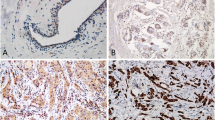
We found that p120{ctn} expression was increased after transfection and the binding capacity of P 120{ctn} to E-cadherin was enhanced. Tyrosine phosphorylation of p 120{ctn} increased after transfection and EGF treatment. p 120{ctn} and β-catenin cellular localization displayd a membrane and cytoplasmic expression pattern, but they translocated into the nucleus for relocalization after p 120{ctn} overexpression plus EGF stimulation. Cell adhesion ability was increased and migration ability reduced after transfection without EGF. Following transfection without EGF cellular proliferation was reduced, but increased after EGF treatment.
Conclusion
Our results suggest that p 120ctn plays an important role in hepatocellular carcinoma cell adhesion, migration and proliferation. In addition there is a relationship between p 120ctn and β-catenin subcellular localization and signaling.
Similar content being viewed by others
References
Reynolds AB, Roesel DJ, Kanner SB, et al. Transformationspecific tyrosine phosphorylation of a novel cellular protein in chicken cell expressing oncogenic variants of the avian cellular src gene. Mol Cell Biol. 1989; 9:629–638.
Downing JR, Reynolds AB. PDGF, CSF-1, and EGF induce tyrosine phosphorylation of p l20, a pp60src transformation associated substrate. Oncogene. 1991; 6:607–613.
Reynolds AB, Daniel J, McCrea PD, et al. Identification of a new catenin:the tyrosine kinase substrate pl20cas associate with E-cadherin complexes. Mol Cell Biol. 1994; 14:8333–8342.
Ohkubo T, Ozawa M. P120ctn binds to the membrane-proximal region of the E-cadherin cytoplasmic domain and is involved in modulation of adhesion activity. J Biol Chem. 1999; 274:21409–21415.
Paulson AF, Fang X, Ji H, et al. Misexpression of the catenin pl20 1A perturbs xenopus gastrulation but does not elicit wnt-directed axis specification. Dev Biol. 1999; 207: 350–363.
Nakopoulou L, Lazaris ACh, Boletis IN, et al. Evaluation of E -cadherin/catenin complex in primary and secondary glomerulonephritis. Am J Kidney Dis. 2002; 39:469–474.
Daniel JM, Reynolds AB. The catenin pl20ctn interacts with Kaiso, a novel BTB/POZ domain zinc finger transcription factor. Mol Cell Biol. 1999; 19:3614–3623.
Ferber A, Yaen C, Sarmiento E, et al. An octapeptide in the juxtamembrane domain of VE -cadherin is important for pl20ctn binding and cell proliferation. Exp Cell Res. 2002; 274:35–44.
Jin DY, Li MF, Hou YD, et al. A guide in molecular cloning. 2nd ed. Beijing: The Sciences Press. 1998;24–26.
Hazan RB, Norton L. The epidermal growth factor receptor modulates the interaction of E-cadherin with the actin cytoskeleton. J Biol Chem. 1998; 273:9078–9084.
Takahashi M, Ikeda U, Masuyama JI, et al. Involenment of adhesion molecules in human monocyte adhesion to and transmigration through endothelial cells in vitro. Atherosclcrosis. 1994; 108:73–81.
Savani RC, Wang C, Yang B, et al. Migration of bovine aortic smooth muscle cells after wound injury: The role of hyaluronan and RHAMM. J Clic Invest. 1995; 95:1158–1168.
Anastasiadis PZ, Reynolds AB. The pl20 catenin family: complex roles in adhesion, signaling and cancer. J Cell Sci. 2000; 113:1319–1334.
Johnson KJ, Boekelheide K. Dynamic testicular adhesion junctions are immunologically unique. I. Localization of pl20 catenin in rat testis. Biol Reprod. 2002; 66:983–991.
Aho S, Levansuo L, Montonen O, et al. Specific sequences in pl20ctn determine subcellular distribution of its multiple isoforms involved in cellular adhesion of normal and malignant epithelial cells. J Cell Sci 2002; 115:1391–1402.
Montanen O, Aho M, Uitto J, et al. Tissue distribution and cell type-specific expression of pl20ctn isoforms. J Histochem Cytochem 2001; 49: 1487–1496.
Nong CZ, Guo LX, Huang HY. Expression of pl20 catenin in the cell lines and tissues of normal liver and hepatocellular carcinoma. Acta Sichuan Univ Med Ed 2003; 34:80–83.
Hengel JV, Vanhoenacker P, Staes K, et al. Nuclear localization of the p 120ctn armadillo-like catenin is counteracted by a nuclear export signal and by E -cadherin expression. Proc Natl Acad Sci USA 1999; 96:7980–7985.
Calautti E, Grossi M, Mammucari C, et al. Fyn tyrosine kinase is a downstream mediator of Rho/PRK2 function in keratinocyte cell-cell adhesion. J Cell Biol 2002; 156:137–148.
Horikawa K, Takeichi M. Requirement of the juxtamembrane domain of the cadherin cytoplasmic tail for morphogenetic cell rearrangement during myotome development. J Cell Biol 2001; 155:1297–1306.
Anastasiadis PZ, Reynolds AB. Regulation of Rho GTPases by pl20-catenin. Curr Opin Cell Biol 2001; 13:604–610.
Prokhortchouk A, Hendrich B, Jorgensen H. The p l20 catenin partner Kaiso is a DNA methylation-dependent transcriptional repressor. Genes Dev. 2001; 15: 1613–1618.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the National Natural Science Foundation of China (No. 30160096).
About this article
Cite this article
Huang, H., Nong, C., He, W. et al. p120 catenin translocation is involved in enhancement of hepatoma cellular malignant features. Chin. J. Clin. Oncol. 2, 693–699 (2005). https://doi.org/10.1007/BF02819534
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02819534